All Issues
Asthma patients with specific genotypes identified for fish oil treatment trial
Publication Information
California Agriculture 65(3):112-117. https://doi.org/10.3733/ca.v065n03p112
Published July 01, 2011
PDF | Citation | Permissions
Abstract
The lifetime prevalence of asthma in California is nearly 20%, and better therapies are needed to manage this common chronic disease. Fish oils containing omega-3 fatty acids are considered a potential therapy for asthma and other inflammatory diseases. Fish oil inhibits the production of arachidonic acid 5-lipoxygenase (ALOX5), an enzyme that exacerbates the lung inflammation that causes asthma. We discuss the genetics of asthma and our preliminary results using a strategy to identify the subgroup of patients who may respond well to treatment with fish oil. These findings, and others, suggest that certain gene polymorphisms of the ALOX5 gene predispose patients to the increased production of inflammatory leukotrienes. Our clinical trials will test the hypothesis that patients with moderate to severe asthma, and with specific high-risk ALOX5 gene sequence variations, will have fewer asthma symptoms when treated with fish oil. The strategy is to decrease the total burden of leukotriene production by supplementing with omega-3 polyunsaturated fatty acids. These studies will also help determine whether genotyping or metabolic profiling (for example, with exhaled breath condensate) can help establish “personalized medicine” for asthma.
Full text
Clockwise from top left, sardines, fish oil pills, flax seeds and soybeans contain omega-3 fatty acids. The omega 3s inhibit the activity of ALOX5, an enzyme that exacerbates lung inflammation and causes asthma.
Asthma is a chronic inflammatory disorder of the body's lungs that is associated with airway reactivity (or “twitchy” airways) and obstruction. It affects an estimated 25 million people in the United States, including 6 million children. Among adults, asthma accounts for approximately 14 million doctor visits and 2 million emergency room visits annually. The annual cost of asthma nationwide is nearly $15 billion, mostly from hospitalizations and emergency room visits but also the cumulative time lost from school absences and missed work (CDC 2002). Sixty to 80% of the health care dollars dedicated to asthma are spent on caring for patients with difficult-to-control asthma, a subgroup that comprises about 10% of all asthmatics (McFadden and Hejal 2000).
In California, as in much of the country, the prevalence of asthma in children and adults has increased dramatically over the past three decades, straining health system resources. Asthma is a classic disease with “gene-environment” interactions, meaning that there is both a host of genes that are associated with the risk of developing asthma but environmental triggers affect this risk. Asthma triggers in the Central Valley of California, for example, include trees, grasses, pollens, air pollution, ozone and possibly insecticides. Scientists do not know all asthma triggers, and it is still difficult to fully explain the increase in asthma in recent decades.
Approximately 13% of Californians have been diagnosed with asthma, and the chance of developing asthma anytime in life is close to 20% (Meng et al. 2003). There are remarkable disparities in asthma prevalence among different ethnic groups in California: among 5 to 17 year olds, the rates are 27.2% for black, 15.6% for Latino and 18.7% for white children. Emergency room visits for asthma in California are four times higher for blacks than whites (144.5 compared to 35.7 visits per 10,000 residents, respectively). The risk of death is highest for adult black women over age 65 (CDPH 2008).
In 2008, the California Department of Public Health (CDPH) published the Strategic Plan for Asthma in California: 2008–2012 (CDPH 2008). The document identified the following focus areas: eliminating asthma disparities among ethnic groups, providing asthma education to all Californians, improving asthma care for all ages and promoting effective asthma management programs. One such program is the UC Davis Asthma Network (UCAN), which includes referral clinics for over 700 patients with difficult-to-control asthma (more than 70% women) (Mealey et al. 2007). Central Valley patients enrolled in this program undergo a rigorous evaluation for environmental and air pollution triggers of their asthma attacks. Other asthma triggers — including indoor house dust mite, cockroach and food allergies — are also determined; the basis of asthma treatment is appropriate avoidance of predictable asthma attacks, making the process of trigger identification critical.
In addition to the CDPH plan, several national and international societies have published asthma guidelines for managing mild to severe asthma, including the British Thoracic Society, Global Initiative for Asthma, and the National Institutes of Health National Asthma Education and Prevention Program (NAEPP). The most recent NAEPP expert panel report (NIH 2007) emphasizes four essential components of asthma care: assessment and monitoring, patient education, control of environmental triggers that affect a patient's asthma, and medications. It provides a detailed approach to achieving asthma control.
Treating asthma
There are two major classes of medications used to treat asthma: inhaled corticosteroids and bronchodilators. Daily inhaled corticosteroids (ICS), such as fluticasone and budesonide, are the preferred long-term asthma control therapy for all ages. Albuterol and other short-acting bronchodilators are used for sudden symptoms of wheezing and shortness of breath, while long-acting bronchodilating beta-2 agonists (LABA), including salmeterol and formoterol, are preferred as add-on maintenance therapy to ICS. Both short- and long-acting beta-2 agonists cause bronchodilation and smooth muscle relaxation, and are commonly prescribed to asthmatic patients for symptom relief. However, LABAs have received black-box warnings from the U.S. Food and Drug Administration (FDA) because a few clinical studies have suggested an increased risk of asthma deaths in patients on these medications. Discussions are ongoing about their use. High-dose ICS and LABA with or without oral, pill-form corticosteroids are also used to treat severe asthma. In addition to medications, patient education is encouraged at all points of asthma care, including clinics, emergency departments and hospitals, pharmacies, schools, community centers and patients’ homes.
Despite the availability of the excellent NAEPP guidelines for physicians, all patients respond differently to treatment, necessitating individualized treatment plans. Some patients have greater control of asthma symptoms with ICS than others; this may be due to differences in the inflammatory cells present in the lungs of asthma patients. Alternatives to inhaled and oral steroids are sometimes needed to help certain patients with severe asthma. While a stepwise approach enables doctors to refine treatments based on individual patient responses and asthma severity, scientists are now developing powerful technologies that may someday enable doctors to identify much more precisely which treatment plans are most likely to yield good results based on a patient's genetic profile.
Glossary
Allele: One of two or more alternative forms of a gene at a specific location on a chromosome.
Eosinophil: A type of white blood cell involved in asthma, allergic responses and parasitic infections.
Genotype: Genetic composition of an organism; set of genes inherited from each parent that determines specific traits.
Leukotriene: Fatty molecule derived from arachidonic acid and involved in mediating inflammation and bronchoconstriction in asthma.
Nutraceutical: A food or dietary supplement that provides medicinal benefits as well as nutritional value.
Phenotype: Trait of an organism that is observable, the result of gene expression and environment.
Polymorphism: Occurrence of two or more phenotypes within a population.
Single-nucleotide polymorphism: A variation of a single nucleotide (A, T, C or G) within a DNA sequence that results in alternative forms of a gene in different individuals.
Asthma affects about 25 million people each year, including 6 million children. The prevalence of asthma is higher among black children and youths (27.2%) than among Latino (15.6%) or white (18.7%) children and youths.
Personalized medicine for asthma
Disease heterogeneity and highly variable patient responses to drugs make prescribing effective treatments difficult for some asthmatics. One approach or set of guidelines does not fit all. The concept of personalized medicine has centered on pharmacogenomics, which recognizes that the variability of patient responses to medical therapy is attributable to genetic variations. Ultimately, physicians hope to prescribe asthma medications based on a clear understanding of which drug each patient may benefit from. However, despite stepwise guidelines designed to tailor asthma therapy to patients based on their disease severity, a personalized approach to asthma management has not been achieved.
Genomics is the study of genes and their effects on a particular disease state. More than 100 genes have been associated with the development of asthma, but fewer than 10 have been identified in more than 10 studies. Most of these genes confer an increased susceptibility to allergic inflammation or hyperresponsiveness of airways.
Genotyping is the analysis of a person's specific gene sequence. Differences in the gene sequences that code for certain proteins offer possible insights into subtypes of asthma; this is a promising approach to personalizing asthma care. From a therapeutic perspective, there is also interest in understanding how diet and nutraceutical supplements influence the development of asthma. Epidemiological data has shown that a Western diet rich in fats and depleted of whole grains correlates with high incidences of asthma, and also that populations that eat large amounts of fish have low incidence rates (Hodge et al. 1996).
Genetic variation and asthma responses
Single nucleotide polymorphisms (SNPs) are variations in the coding of a DNA sequence that occur as a result of a single nucleotide change. They are the most frequent type of genetic variation, with about 10 million identified in the human genome. One such SNP encodes for the receptor ADRB2. This receptor is important in human health, and its different forms, point mutations or down-regulation are associated with nocturnal asthma, obesity and type 2 diabetes.
For asthma sufferers, this genetic variation (a switch from guanine to adenine at one position) may be associated with a worse response to asthma inhalers such as albuterol or salmeterol beta-2 agonists (substances that initiate a physiological response when combined with a receptor). Because human cells have two sets of chromosomes, there are three possible genotypes that result from this SNP (A/A, A/G and G/G). Asthmatics with the A/A type have shown poorer improvements in expiratory flow rates after receiving albuterol than asthmatics with either G/G or A/G genotypes.
We used a genomic approach to test whether fish oil supplements could improve clinical outcomes for people with difficult-to-control asthma. Fish oil concentrate contains omega-3 polyunsaturated fatty acids (n-3 PUFAs) — comprised of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) — which inhibit the activity of arachidonic acid 5-lipoxygenase (ALOX5). ALOX5 is an enzyme present in many inflammatory cells that makes inflammatory proteins called leukotrienes. Leukotriene levels are very high in asthma patients and contribute to the inflammation and airway reactivity that are the hallmarks of the disease. ALOX5 is an important target for newer drugs being developed for asthma. Our clinical trial is studying whether patients with moderate to severe asthma who have specific high-risk ALOX5 gene polymorphisms have fewer asthma attacks or exacerbations when using a fish oil supplement.
Asthma genomics
Genetic studies in asthma are difficult to interpret because of the sheer number of gene abnormalities that correlate positively with the presence of asthma. However, of more than 100 genes of potential interest, ADRB2 and ALOX5 are perhaps the most clinically relevant.
ADRB2.
ADRB2 is the gene for the beta-2 adrenergic receptor (Warrier and Khurana Hershey 2008). It is of interest to clinicians because there is good evidence that the variability in patient responses to beta-2 agonists such as albuterol can be attributed to single nucleotide polymorphisms (SNPs) on the ADRB2 gene (see box) (Moffatt 2008). An SNP is a DNA sequence variation of a single nucleotide that can lead to very different protein structure or function. It is important that doctors and scientists understand these SNP variations in order to determine whether certain medications will affect patients differently. Certain ADRB2 SNPs predict poor response to beta-2 agonist medications. This new knowledge contributed to the FDA's decision to place a black-box warning on LABAs. Because there are risk factors with medical treatment, utilizing SNP information could eliminate trial and error in treatment. This is an example of how pharmacogenomics may provide doctors and scientists with the information to more appropriately target medications to certain patients.
ALOX5.
ALOX5 encodes the enzyme that produces leukotrienes, which play a role in the predisposition to severe asthma. It may be the second most clinically important gene discovery in asthma. Polymorphisms in ALOX5 and also in 5-lipoxygenase-activating-protein (FLAP) are associated with the excessive production of leukotrienes (Koshino et al. 1999). The levels of ALOX5 and FLAP messenger RNA — the RNA molecule that is the blueprint for the protein — are increased in peripheral blood leukocytes from asthmatics (Koshino et al. 1998). In addition, the concentrations of leukotrienes in sputum were greater in patients with asthma than in nonasthmatic subjects (Ogawa and Calhoun 2006).
A study by Klotsman et al. (2007) found that a subset of asthmatics with ALOX5 polymorphisms were predisposed to having higher cysteinyl-leukotriene (cys-LT) concentrations, and they constituted an asthma phenotype more likely to respond to a leukotriene-inhibiting drug such as montelukast. Another study examined the association of ALOX5 promoter genotypes with responses to montelukast therapy. While on therapy, patients with the ALOX5 SNP variant had fewer asthma symptoms than controls (Lima et al. 2006).
Fish oil and the leukotriene pathway
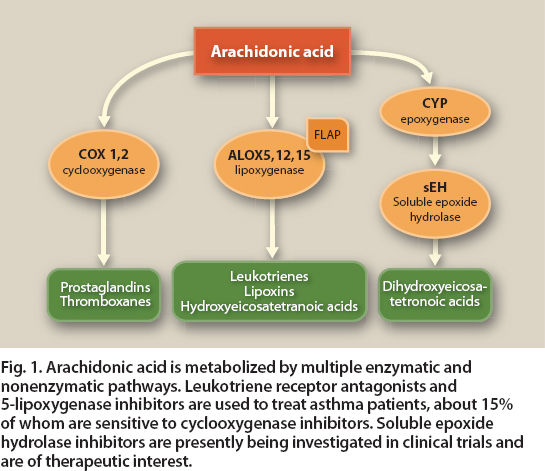
Some asthma patients benefit greatly from treatment with anti-leukotriene drugs, which can attenuate the effects of leukotrienes not blocked by steroids. Overall, however, the anti-leukotriene drugs are only moderately anti-inflammatory and are considered second-line asthma therapies, after inhaled corticosteroids (Tantisira and Drazen 2009). The leukotriene inhibitors (montelukast and zafirlukast) and 5-lipoxygenase inhibitor (zileuton) improve airway function primarily by blocking the inflammatory effects of various leukotrienes (fig. 1).
Fig. 1. Arachidonic acid is metabolized by multiple enzymatic and nonenzymatic pathways. Leukotriene receptor antagonists and 5-lipoxygenase inhibitors are used to treat asthma patients, about 15% of whom are sensitive to cyclooxygenase inhibitors. Soluble epoxide hydrolase inhibitors are presently being investigated in clinical trials and are of therapeutic interest.
Nutraceuticals such as fish oil may play a role in the treatment of asthma by inhibiting 5-lipoxygenase, reducing the need for medicines. Components in fish oil (omega-3 fatty acids, composed of EPA and DHA) suppress the production of arachidonic acid–derived leukotrienes via the ALOX5 pathway. Arachidonic acid is an omega-6 fatty acid present in the body's cell membranes; it is a key substrate for the synthesis of eicosanoids, potent mediators of the inflammatory response that include cys-LTs, prostaglandins and thromboxanes (Calder 2006).
Byproducts of these mediators (LTC4 and related cyst-LTs) are directly involved in the bronchoconstriction and increased mucus production seen in the airways of asthmatics. EPA-enriched fish oils may competitively inhibit the production of LTC4 by competing with arachidonic acid as a substrate for ALOX5 (fig. 2). EPA may also suppress an allergic response in asthma by competing with arachidonic acid as a substrate for the production of leukotrienes.
Fig. 2. Specifically enriched fish oils compete with arachidonic acid for the ALOX5 enzyme, potentially decreasing the production of inflammatory leukotrienes. We hypothesize that asthma patients with specific high-risk ALOX5 gene polymorphisms will derive greater clinical benefit from fish oils enriched with eicosapentaenoic acid (EPA), an omega-3 fatty acid.
One leukotriene, prostaglandin E2, mediates the formulation of immunoglobulin E (IgE), an antibody responsible for initiating allergic responses. It is usually elevated in patients with asthma. EPA and DHA are precursors to potent bioactive mediators that possess both anti-inflammatory and protective properties.
The World Health Organization recommends a dietary ratio of omega-6 to omega-3 fatty acids of four to one. Studies have shown that after just 2 weeks of dietary supplementation with EPA, the composition of cell membranes can change to include higher levels of omega-3s (Wong 2005). Changing the membrane composition of fatty acids via daily fish oil intake may modulate the production of inflammatory leukotrienes and have a significant impact on curbing asthma symptoms.
We are performing a clinical study to determine whether dietary supplementation with EPA- and DHA-enriched omega-3 fatty acids can ameliorate the production of inflammatory leukotrienes and decrease the number of acute exacerbations in patients with moderate to severe asthma. The hypothesis is that fish oil supplementation can impede the production of inflammatory leukotrienes in asthmatics with SNPs in the promoter of ALOX5. Furthermore, we believe that this subset of asthmatics, which we term high risk, is likely to have one or more polymorphisms of specific genes in the 5-lipoxygenase pathway.
This new approach — using genotyping to identify a subset of patients with asthma who are likely to respond to a specific therapy — can have far-reaching implications in terms of asthma outcomes. Self-administration and ingestion of fish oils as anti-inflammatory or high-cholesterol medications is common in the general population; several people considered for our study were unwilling to discontinue their home supplements and were therefore ineligible. Subjects interested in enrolling had a 2-month fish oil–free “washout” period. Fish intake was limited to less than three servings per week, and a dietary questionnaire was given to assess recent fish consumption. In this manner, we hoped to standardize extraneous fish oil intake among the study subjects.
Genotyping of asthmatics
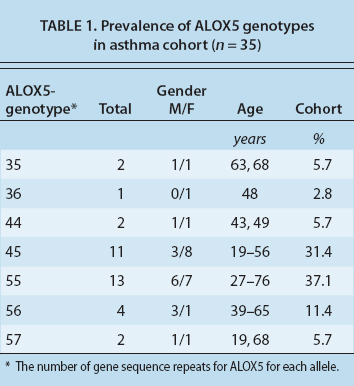
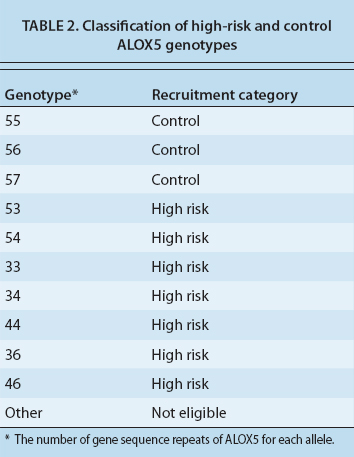
We have performed genotyping of the ALOX5 gene in a full cohort of 35 subjects, 22 of whom are female. The mean age is 46.7 years with a range of 19 to 76 years, reflecting the diversity of moderate to severe asthma in adults (table 1, page 109). Subjects were identified as having a low-risk (control) genotype, or a high-risk genotype based on the number of gene sequence repeats (three to seven) for the binding site of the transcription factor Sp1, which regulates the expression of ALOX5. The most common DNA sequence, or allele, has five such repeats, and a 55 genotype reflects the presence of five repeats on each of the two DNA strands (table 2).
Our preliminary studies suggest that subjects with non-five repeat alleles have more severe asthma and may respond better to omega-3 fatty acids. We classified the people with 55, 56 and 57 sequences as the control and subjects with either three or four repeats on at least one DNA sequence as high risk and more likely to respond to fish oil. Among our 35 subjects, 16 (45.7%) were high risk. We hypothesize that this group will ultimately show more improvement in their asthma with fish oils than the control group.
We had a much higher percentage of subjects with the high-risk ALOX5 genotypes than reported in the general population, and more than we had predicted. Less than 50 subjects were therefore screened. Overall, this finding suggests that the prevalence of these genotypes is higher in carefully characterized cohorts of severe, persistent asthma patients.
Can exhaled breath help?
Pharmacogenomics and personalized medicine have already been applied in the field of oncology; several anticancer drugs have been shown to be beneficial to patients with certain gene mutations. One problem with this approach is that numerous genes (more than 100 in the case of asthma) are potential candidates for targeting in a single disease, and it is not feasible to test all new drugs in prospective studies that focus on single genes, such as ours. Instead, determining responses to therapy may be best served by analyzing proteins and their metabolic byproducts as biomarkers in blood serum and other body fluids.
Patients with moderate to severe asthma often have persistent symptoms, even with use of inhaled corticosteroids. There is evidence that a specific genetic variation may be associated with poor responses to some medications.
The lung is easily sampled, which is an advantage for studying lung diseases such as asthma. For example, Brasier et al. (2008) were able to sample lavage fluid from the lungs of patients with severe and mild asthma. They identified 10 cytokines, molecules involved in cell signaling, that could be used to characterize several asthma subtypes. These studies offer potential new diagnostic tools that could be used to identify subtypes of asthmatics, monitor disease severity and target therapies to specific groups of patients who will respond to them.
However, obtaining lung lavage fluid is fairly invasive. Less invasive methods include the measurement of eosinophil numbers in sputum and, more recently, the concentration of exhaled nitric oxide in breath (Ueno et al. 2008). The fractional excretion of nitric oxide has a reported specificity of up to 90% for the diagnosis of asthma (Kharitonov and Barnes 2006). In addition to nitric oxide, there are more than 20 biomarkers that are increased in the liquid condensate of exhaled breath of asthmatics, including leukotrienes and other inflammatory proteins.
Having a broader profile of measured biomarkers would provide a more accurate assessment of a patient's disease severity. Exhaled breath condensate (EBC) analysis has emerged as a promising method to noninvasively measure the concentration of inflammatory mediators. EBC consists of volatile and nonvolatile components, which are evaporated from fluid in the airway epithelial lining. The collection of EBC is performed by cooling exhaled air during spontaneous respiration (Kostikas et al. 2008). Several studies have shown that patients with moderate to severe asthma have significantly higher concentrations of leukotrienes in EBC than mild asthmatics and controls (Samitas et al. 2009).
Metabolomic analysis identifies potential biomarkers for a disease by analyzing the products of cell metabolism using mass spectrometry or high-resolution proton nuclear magnetic resonance spectroscopy (1H-NMR). All the molecules that produce a signal in the mass or NMR spectrum are detected, creating a “metabolic fingerprint” of the analyzed sample. Metabolomics have been used to study inborn errors of metabolism. This method can be applied to EBC, enabling the detection of known metabolites and the prediction of novel biomarkers that would allow further characterization of asthma phenotypes and the prediction of drug efficacy. For example, Baraldi et al. (2009) used metabolomic analysis of EBC to demonstrate a difference between biomarkers found in the airways of asthmatic and healthy children.
Toward effective asthma treatment
This clinical study addresses the need to focus on patients with more severe asthma, as well as patients with specific genotypes or metabolomic phenotypes that may identify them as responsive or nonresponsive to therapy. Preliminary data from ours and other studies suggests that patients with certain polymorphisms of the ALOX5 gene are more susceptible to the production of inflammatory leukotrienes. This minority of asthmatics likely represents the fraction of asthmatics that would improve significantly from anti-leukotriene drugs, including a 5-lipoxygenase inhibitor or leukotriene receptor antagonist. These drugs are not completely efficacious in blocking this pathway, however, and a strategy of decreasing the total burden of leukotriene production further by supplementing with omega-3-rich fish oil is rational.
Many genes are associated with the risk of developing asthma; environmental triggers such as air pollution compound the risk.
Prior clinical trials focused primarily on subjects with milder asthma. Patients with moderate to severe asthma are likely to notice a significant benefit from an intervention such as fish oil. These patients often have persistent airflow obstruction, despite therapy with inhaled corticosteroids. Our trials involving such patients will conclude this year. Further trials are needed to assess whether fish oil supplements will be effective in asthma patients receiving standard medications.