All Issues
Salmonella in sewage effluent raises ecological and food-safety concerns
Publication Information
California Agriculture 54(5):62-68. https://doi.org/10.3733/ca.v054n05p62
Published September 01, 2000
PDF | Citation | Permissions
Abstract
Salmonella bacteria continue to be an important public-health problem and have a serious economic impact on the U.S. poultry industry. Although the majority of human infections traceable to Salmonella enteritidis are related to egg consumption, this serotype has also been isolated from a variety of nonegg food items such as meat, vegetables and fish. In Southern California, S. enteritidis phage type 4 infection is the predominant serotype found in human patients. However, the organism has also been found in municipal sewage effluent and in rodent and poultry environments. S. enteritidis phage type 4 was first detected in human patients in Southern California in 1990, but wasn't isolated from food-producing animals until its occurrence at a Southern California egg-layer ranch in 1994. An epidemiological study revealed that the layer flock was infected via effluent originating from a nearby municipal sewage-treatment plant. Human sewage effluent was the primary environmental source, combined with wildlife that further amplified and disseminated the bacteria. This discovery has important implications for understanding the ecology of S. enteritidis infection in poultry and humans and developing appropriate methods to prevent its further spread.
Full text
Zoo animals have been a source of Salmonella infection for humans and other animals. Additional sources include eggs, contaminated water and a variety of foods, such as vegetables, fish, poultry meat and dairy products. The U.S. poultry industry is spending $10 million to $20 million to control Salmonella enteritidis.
Salmonellosis is a major public-health problem and continues to have a serious economic impact on the U.S. poultry industry (Morales and McDowell 1999). The estimated cost of the Salmonella enteritidis-control program to the poultry industry nationwide is $10 million to $20 million.
Of some 2,400 known serotypes of Salmonella, Salmonella enterica or (S. enteritidis) is of particular concern. Widely present in the environment and in the intestinal tracts of rodents, Salmonella is often associated with clinical disease in humans but not necessarily in the host animal. Because humans are constantly modifying the environment, the Salmonella balance in animals and humans is dynamic and often in flux.
The global increase in S. enteritidis infections in humans and poultry suggests a new pandemic of the disease. S. enteritidis isolates increased in 24 of 35 countries (69%) between 1979 and 1987 in North America, South America, Europe and Africa (Rodrigue et al. 1990; Altekruse and Swerdlow 1996).
Between 1985 and 1995, the first 10 years S. enteritidis was recognized in the United States, the infection spread from the Northeast to the rest of the country. In the United States, S. enteritidis comprised 25% of all Salmonella isolated by 1995, compared with 5% in 1985 (Gomez et al. 1997). The rate of S. enteritidis-related illness reported to the Centers for Disease Control and Prevention (CDC) increased from 0.6 per 100,000 population in 1976 to 3.6 per 100,000 in 1996. From 1996 to 1998, the rate of confirmed S. enteritidis cases declined to 2.2 per 100,000. However, outbreaks continue to occur. For example, there were 44 confirmed outbreaks of S. enteritidis infections reported in 1999 in the United States; of the 19 outbreaks in 1999 for which a vehicle could be confirmed, 15 (79%) were associated with shell eggs (CDC 2000).
It has been suggested that the worldwide increase of S. enteritidis infections in humans is traceable to modern poultry-farming practices, combined with the decline in the genetic diversity of domestic fowl (Pignato et al. 1996). However, this hypothesis does not explain why human infections with Salmonella strains other than S. enteritidis, also carried by poultry, have not increased during the same period (Rodrigue et al. 1990).
Baumler et al. (2000) hypothesize that the emergence of S. enteritidis was the result of this Salmonella serotype filling the ecological niche vacated by two poultry host-specific pathogens, S. pullorum and S. gallinarum. Both of these poultry pathogens were successfully eliminated through implementation of the 1935 National Poultry Improvement Plan, which was designed to reduce pullorum disease (bacillary white diarrhea) caused by S. pullorum. The plan was revised in 1954 to also address fowl typhoid, an illness caused by S. gallinarum (Bullis 1977).
A technician places a Moore swab in sewage effluent. The swab traps particulate matter and bacteria such as Salmonella. The swab is attached to a string or wire and anchored to the creek bank. The swab is kept 4 to 7 days in the effluent as a sensitive means of finding Salmonella, then transported to the laboratory for analysis.
Once these two poultry pathogens were eliminated, the theory is that S. enteritidis gained a foothold in poultry flocks (Baumler et al. 2000). Chickens infected with either S. pullorum or S. gallinarum may have been inadvertently cross-protected against infection with S. enteritidis due to the similarity of the O12 and O9 antigens found in all these organisms. According to this theory, in the past, poultry flocks were naturally immunized with the antigens against S. enteritidis.
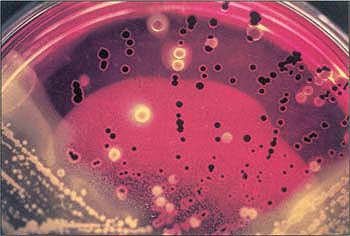
Numerous black colonies growing on XLT4 agar plate (special selective media) suggest Salmonella organisms. The suspect black colonies undergo further testing and are confirmed to be Salmonella.
However, the Baumler hypothesis does not explain why turkey flocks in most developed countries that were once affected by pullorum/typhoid disease and are now clear of it do not have S. enteritidis infection. This hypothesis also overlooks that in many regions of the world, such as in South and Central America, the Middle East, the Indian subcontinent and parts of Africa where pullorum and typhoid are prevalent, infections did not restrain the emergence and spread of S. enteritidis (Pomeroy and Nagaraja 1991; Silva 1985).
Uninfected chicken with abdominal and thoracic organs exposed. Note the smooth and glistening surfaces of the organs.
Infectious symptoms and sources
Salmonellosis due to S. enteritidis usually presents as a self-limited diarrhea with abdominal pain, fever and occasionally vomiting. Pregnant women, elderly persons and those who are immunocompromised are at increased risk for the development of invasive disease (Passaro et al. 1996; Steinert et al. 1990).
Although the majority of S. enteritidis infections in humans are traced to egg consumption (St. Louis 1988), S. enteritidis has also been isolated from a wide variety of nonegg food items including poultry meat, dairy products such as milk/cheese, fish (tuna and salmon), vegetables, tomato sauce, lamb cutlets and eggless cake (Scuderi et al. 1996).
In one study, S. enteritidis isolates constituted 13% to 77% of all serotypes captured from people who handle food (Thiel 1999). During home investigations of children infected with Salmonella, identical types were identified among patients in multiple locations and from several food items in the same home. This finding underscores the importance of contagion within the home in the ecology of salmonellosis among people (Schutze et al. 1999).
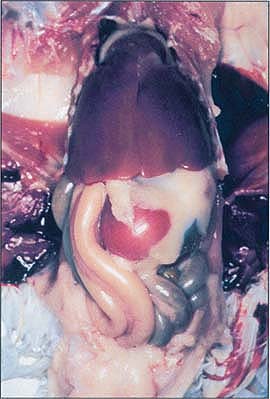
Chicken organs affected by Salmonella enteritidis. Note the fibrinous layer on pericardial sac, liver and the thickened yolk material between the intestinal loops, and a soft-shell egg free in the abdominal cavity.
Zoo animals.
Zoo animals also often carry Salmonella and can be a source of infection for other animals and humans. Zoos, with their concentrated assemblies of exotic and native animals, can harbor predominant strains associated with animal feeds. A recent outbreak of S. enteritidis in people implicated the Komodo dragons as carriers at a metropolitan zoo (Friedman et al. 1998). People became infected with S. enteritidis by touching the contaminated barrier between the dragons and the visitors, most likely followed by some manner of oral inoculation such as consuming food.
Southern California isolates
Phages or bacteriophages are viruses that infect susceptible host bacteria. There are more than 50 phage types of S. enteritidis (Humphrey 1999). S. enteritidis strains most commonly associated with human outbreaks in the United States belong to phage types 8, 13 and 13a (Altekruse and Swerdlow 1996; Mishu et al. 1994).
However, a Southern California study of sporadic human cases showed that 95% (24 of 26 randomly selected isolates) were S. enteritidis phage type 4 (Passaro et al. 1996). In contrast, S. enteritidis phage type 4 is not common among Northern California patients (Passaro et al. 1996).
The reason for the disparity is not known, but we speculate that the high population densities characteristic of Southern California, in combination with the large number of international tourists, may have resulted in the introduction and spread of S. enteritidis phage type 4 among humans. Furthermore, the climate in Southern California tends to be warmer than in the northern part of the state, possibly resulting in enhanced food-safety risk. Warmer climates invite outdoor eating either at commercial establishments or at picnics, exposing people to foods that may be improperly prepared or stored.
The first recognized human outbreak of S. enteritidis phage type 4 in the United States that was not associated with foreign travel occurred in Texas in 1993 (Boyce et al. 1996). This date is important because before 1993 S. enteritidis phage type 4 had not been identified in food-producing animals in the United States.
S. enteritidis phage type 4 was first detected in humans in California in 1990 (Passaro et al. 1996), 4 years before its discovery in food-producing animals. Since 1990, S. enteritidis phage type 4 has continued to be isolated in Californians, with important implications for how this bacterial pathogen may have spread to food-producing animals.
The creek and the poultry ranch
The appearance of S. enteritidis phage type 4 in a commercial flock in May 1994 in Southern California raised serious concerns within the poultry industry and the U.S. Department of Agriculture (Kinde et al. 1996b). Epidemiological investigation revealed that a creek located about 164 feet from the infected poultry ranch was the source of S. enteritidis phage type 4 for the infected layer flock. The effluent from a sewage treatment plant about three-fifths of a mile upstream comprised the creek's entire water supply. The treated sewage effluent in the creek was cloudy and foamy, occasionally producing a fecal odor, which suggests inadequate treatment allowed harmful bacteria to pass through.
More importantly, isolates of S. enteritidis phage type 4, each with identical molecular markers (plasmid profiles and restriction endonuclease analysis), were cultured from the creek effluent, internal organs and eggs of chickens, and wild mice inhabiting the area. The isolation of S. enteritidis phage type 4 from wild mice suggests that these rodents may have been the biological vector, effectively spreading the bacteria from the effluent to the layer flock.
The creek provided a likely source of infection for these rodents, either through water ingestion or direct contact, or indirectly if the effluent then contaminated seeds or other food eaten by the mice. Once infected by the effluent, the mice readily contaminated the feed bins in the layer houses, resulting in the first isolation of S. enteritidis phage type 4 in poultry in California (Kinde et al. 1996a).
This research demonstrates that foodborne pathogens can enter the food chain via human contamination of the environment, subverting the common wisdom that food-producing animals are the primary contaminating source. Use of minimally treated human sludge as fertilizer or soil amendment increases the possibility of spreading Salmonella and other pathogens from humans to agricultural and other domestic environments. Discharging sewage or other effluents that contain infectious pathogens into our waterways can increase the risk of further infection to humans and animals.
In most developed countries, turkey flocks that were once infected by S. pullorum or S. gallinarum are not infected with S. enteritidis.
Other infection sources
Several instances of human and animal salmonellosis have been attributed to environmental pollution with sewage sludge, gulls and slaughter-house effluents (Harbourne et al. 1978.) Kinde et al. (1997) found that effluent from 11 of 12 sewage treatment plants and two of three surface-water sources yielded Salmonella organisms. S. enteritidis phage type 4 was recovered from three of these treatment-plant effluents and, surprisingly, from a potable-water reservoir.
The food chain.
The use of effluents and minimally treated sewage sludge in agriculture, if conducted inappropriately, has the potential to spread Salmonella in the environment. Moreover, if contamination of food-stuffs or agricultural commodities occurs, agricultural workers and their families as well as consumers are at risk of infection (Shuval et al. 1984; Hedberg et al. 1994).
With respect to humans serving as a source of Salmonella, a person with acute salmonellosis can excrete Salmonella in quantities of 108 to 1011 organisms per gram of feces (Brisou 1968; Grunnet 1975). The daily use of water per capita is estimated to be nearly 100 gallons (400 liters) (EPA 2000). Therefore, in a community of 100,000 people with a single active case of salmonellosis, the Salmonella concentration in raw sewage can approximate 10,000 Salmonella per liter of sewage [(109Salmonella/g feces) x (400 g feces/person/day) / (105 people) x (400 liters/person/day) = 104Salmonella/L].
The number of salmonella organisms that cause illness to humans varies depending on factors including the type of food, age of the host, and factors known to affect the immune status of the host such as infection with HIV, leukemia, use of immunosup-pressive drugs and so on. In recent outbreaks, food items containing between 105 and 108 organisms have been found.
Small children playing in effluent, which contains Salmonella and other potentially pathogenic organisms. In the authors' research, a chain of S. enteritidis contamination can be traced from humans to effluent to rodents to poultry, and back to humans.
Waste treatment/effluent.
The extent to which waste-treatment processes reduce Salmonella concentrations depends upon the treatment-plant methods. Nevertheless, small numbers of infective Salmonella are typically present in primary or secondary effluents. If the effluent is cloudy or odorous when discharged into the environment, people tend to refrain from swimming or bathing in the water. However, when the effluent appears clean and has little or no odor, as is often the case, the water is appealing and people will swim in it. Swimming or bathing in effluent is a potential health risk.
We have observed children playing and other people fishing, swimming or bathing in the effluent within 33 yards of an outfall. Even when standard water-quality tests such as fecal coliform counts indicate that the effluent is safe, pathogenic microorganisms can be isolated (Cherry et al. 1972). Therefore, the coliform test may not always be a suitable indicator of the microbiological safety of water.
Rodents and disease.
In the early 1900s, human S. enteritidis infections were associated not with poultry but with rodents, the only known animal reservoir for this pathogen (Edwards and Bruner 1943). Rodents, especially mice and rats, are frequent visitors to poultry houses where they can contaminate feed bins. Since mice feed on their own excrement, once S. enteritidis enters a mouse population, it tends to remain for generations. Consequently, chickens can become infected by eating feed contaminated with feces.
Investigation of an S. enteritidis phage type 4-infected poultry ranch revealed that resident feral animals such as cats, mice and skunks were infected by organisms identical to those found in the chickens (Kinde et al. 1996a). The primary source of infection was traced to sewage effluent from a nearby creek. Twenty-seven different serotypes were identified in the effluent over the course of 6 months, with S. enteritidis the most common.
In another survey of resident feral animals around seven waste-treatment plants, 17% of samples (31 of 178) yielded 18 different serotypes. Again, those same types were simultaneously found in nearby sewage effluents. This suggests that sewage effluent discharged into the environment is serving as the bacterial source of infection for feral animals, which then amplify the infection and disseminate it throughout the local environment, including by infecting food-producing animals (Kinde et al. 1996a).
The chicken or the egg?
This theory of sewage effluent functioning as a primary environmental source of Salmonella, and wildlife acting to amplify and disseminate the bacteria, has important implications for how we understand the ecology of S. enteritidis infection in poultry and humans. In the United States, there are five companies with a total of 800,000 birds that are the primary breeders of the nation's multiplier flocks, which produce the final 230 million commercial layers that generate the nation's egg supply (St. Louis et al. 1988). Given the ubiquitous vertical integration of the layer industry, if poultry — and especially the primary or multiplier flocks — are the primary reservoir for S. enteritidis in this country, one would expect widespread infection with S. enteritidis in layer flocks via vertical transmission from bird to bird.
However, widespread infection is not seen in our layer flocks, suggesting instead that most S. enteritidis infection is due to horizontal transmission. In Southern California, S. enteritidis is rarely isolated in routine surveillance using environmental drag swabs from poultry ranches. When S. enteritidis is isolated, most belong to phage type 4 (Kinde, unpublished data). The reason for the predominance of S. enteritidis phage type 4 in Southern California poultry flocks is not well-known, but we have found that S. enteritidis is well-established in the environment of rodents, surface waters and effluents (Kinde et al. 1996a, 1996b, 1997).
It is likely that these contaminated environments could directly contribute to the incidence of human salmonellosis, either through direct contact with contaminated water or indirectly through biological vectors such as rodents, which can contaminate a variety of human foods.
This problem is a classic “chicken-or-egg (or was it something else?)” hypothesis: Were Southern California poultry infected due to an original human infection → contaminated effluent → infected rodents → infected poultry, or was the Southern California public originally infected through foodborne transmission due to contaminated eggs or a chicken carcass? The published literature suggests the infections began with humans, at least in Southern California.
Future in focus: Prevention strategies
S. enteritidis is a minor problem for poultry, rarely making them clinically ill, but poses a serious concern for food safety along the food-production chain — from the animal-feed supplier to the livestock producer, to the distributor, to the retailer and finally the consumer.
To prevent contamination, animal feed such as grains or forage should be grown using clean water on ground not contaminated with microbial pathogens, and processed and stored in an environment free of vermin and pests. Feed samples should be routinely monitored to ensure that they are free of Salmonella.
There are between 26 million and 32 million laying hens in California, according to the California Egg Commission. The California Egg Quality Assurance Plan (2000) was developed in 1994 to ensure the high quality and safety of eggs. The voluntary program contains 20 core components, which form the basis of a hazard analysis and critical control points (HACCP) plan. The plan calls for the purchase of chicks and pullets from hatcheries participating in the National Poultry Improvement Plan, an effective flock-health program that includes practices such as vaccination, routine mortality profiles, a rodent monitoring and reduction program, and effective biosecurity systems. The vast majority of California egg producers have adopted quality assurance plans.
Mishandled eggs.
A large proportion of foodborne microbial risk from eggs is related to the consumption of eggs that have been cross-contaminated or mishandled from the farm to the table, rather than contamination occurring within the chicken. Food handlers should play a major role in preventing the introduction and spread of S. enteritidis during food processing and preparation.
Basic sanitation practices should be implemented and enforced for food processing and handling, manufacturing controls and personal hygiene. Persons known to be ill or carrying potentially communicable diseases should be excused from the food-handling area. The outside of commercial food establishments and certainly the interior of buildings and facilities should be free of pests and other contamination sources.
Minimizing the risk of human and animal infection with Salmonella remains a central concern for those working in public health, municipal waste and animal agriculture. Control of S. enteritidis, given its ability to be transmitted among humans, rodents and poultry, is particularly challenging due to its presence in the environment. To break the chain of infection that may exist between humans and poultry, a multilevel approach is needed.
Breaking chain of infection.
Wastewater-treatment plants, particularly those that discharge into the environment, must ensure that their effluent is free of Salmonella. This can be achieved by implementing the newer technologies for disinfection such as filtration and UV systems. Effluents should be monitored regularly for adequate disinfection to ensure harmful bacteria are not released in the environment through the sewage effluent.
Efforts must also be improved to minimize direct contact between humans and contaminated effluents, such as fishing or swimming in streams polluted by effluent. A concerted effort must be made to minimize animal infection with human pathogens. Methods of wastewater disinfection are available and in development for improved effectiveness. In many cases disinfection is reasonably effective, but municipal treatment plants should be required to monitor constantly for Salmonella before the final effluent is discharged into the environment. We believe these are key steps in the effort to protect the public from Salmonella and to help ensure that the food supply is free from microbial pathogens.
S. enteritidis-prevention strategies should include using good agricultural practices, implementing egg-safety programs, and ensuring that food is handled properly during processing, cooking and preparation.
In the next 25 years, the population of California is expected to increase significantly. In order for the state to maintain its global competitiveness in agriculture and provide affordable, safe food for its citizens, science-based and risk-based approaches have to be used in the food safety system. Biotechnology has enormous promise and implications for food safety, nutrition and water-quality management. Recent advances in genetic engineering, such as DNA probes, immunoassays and enzymatic tools, can be useful for detecting or monitoring foodborne pathogens in sewage effluents and surface waters. The detection and proper elimination of pathogens such as Salmonella in sewage effluents and the conversion of such potentially contaminated effluent to productive and environmentally sustainable uses will benefit the state enormously.
We need to develop molecular methods to discriminate among S. enteritidis phage type isolates so that trace-back investigations from human illness outbreaks are correctly and effectively characterized and dealt with. The identification and characterization of S. enteritidis-infected flocks based on egg contamination should be one of the priorities of research. This is critical information for the industry to know in order to use its resources appropriately for implementing science-based prevention strategies for reducing S. enteritidis infection in humans without adverse economical impact.