All Issues
Metam-sodium kills beneficial soil fungi as well as cotton pests
Publication Information
California Agriculture 50(5):42-44. https://doi.org/10.3733/ca.v050n05p42
Published September 01, 1996
Abstract
Mycorrhizal fungi colonize cotton roots and stimulate plant growth by absorbing minerals from the soil. These beneficial fungi can be adversely affected by metam-sodium, a soil-applied general biocide frequently used for weed control in cotton. Although metam-sodium also kills disease-causing fungi that reduce cotton stands, the advantages of pest control with metam-sodium must be weighed against added fertilizer costs needed to offset the loss of mycorrhizae.
Full text
Metam-sodium is commonly used to fumigate soil before planting cotton to control weeds and seedling disease, including the damping-off shown here.
Soilborne pathogens potentially reduce seed germination, plant stands and cotton yields. In 1994 the estimated loss in lint yields caused by seed and seedling diseases in California was 48,370 bales (1.7% of total production), despite the availability of chemical seed treatments effective against the major seedling pathogens. Soil-applied metam-sodium, a general chemical biocide, is commonly used on cotton fields in the San Joaquin Valley to control weeds in the seedrow. It has been observed that this method of application, aimed primarily at weed control, sometimes increases plant stands, presumably due to seedling disease control, and occasionally decreases plant growth, possibly due to the disruption of root colonization by beneficial mycorrhizal fungi. The purpose of this study was to determine the effects of metam-sodium on cotton growth and seedling disease in fields with histories of poor stand establishment.
Experiments were conducted in three locations (Madera, Fresno and Kern counties, Sites 1, 2 and 3, respectively) in the San Joaquin Valley. Sites 1 and 2 were on clay loam soils; Site 3 was on a sandy loam soil. Treatments were arranged in a 2-by-3 factorial design to test the effects of seed treatments and metam-sodium on cotton seedling disease, plant growth and mycorrhizal infection. Main-effect comparisons included nontreated seed vs. seed treated with the standard fungicides metalaxyl (Apron) for the control of Pythium-induced seedling disease, and with chloroneb (NuFlow D) for the control of Rhizoctonia solani-induced seedling disease, and metam-sodium (Vapam) applied to the soil at 0, 50, or 100 gallons/acre. Treatments were arranged in a randomized complete block design with six replications. Each experimental unit consisted of one 50-foot row on 38-inch centers. Cotton (Acala SJ-2 at Sites 1 and 2, Acala Maxxa at Site 3) was planted at a rate of 200 seeds per 50 feet.
Metam-sodium was applied to the beds by a CO2-powered injection device built onto an implement sled. The injection device consisted of an 8-inch spray blade outfitted with either a K3.0 or a K2.0 flowjet nozzle placed underneath the spray blade, which was attached to a shank adjusted to deliver the material at a depth of 6 to 8 inches below the surface of the bed. A small covering shoe or a pair of disks were pulled behind the shank to cover and seal the soil after treatment. The applications were made on preformed, preirrigated beds 4 to 6 weeks prior to planting.
Stand counts were recorded 14 or 15 days after planting. Plant growth measurements were taken on 8 to 10 random plants per plot 8 to 12 weeks after planting. Plant growth measurements included plant height, number of nodes, position (node) of the first boll and plant vigor (height/total number of nodes). For plant measurements, the cotyledons were considered node 0 and the reference point for plant height. Yields were determined by hand harvesting 13.8 feet/plot (1 / 1000 of an acre) from each plot after defoliation.
Eight weeks after planting, 10 soil cores were collected at a depth of 0 to 8 inches near the base of the plants in each experimental unit and bulked. Feeder roots were extracted from the soil samples, cut into 0.4-inch-long segments, cleared in potassium hydroxide and stained with trypan blue. Each of 10 to 20 segments per sample was examined under a microscope for the presence of hyphae, vesicles or spores of mycorrhizal fungi. Results were expressed as the average percentage infection in the root segments.
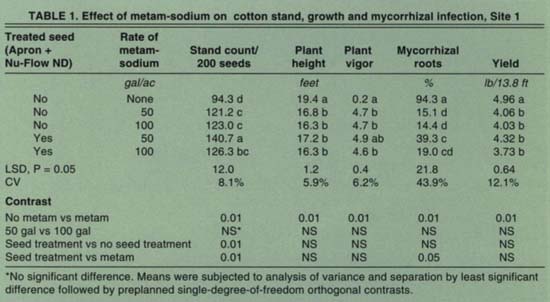
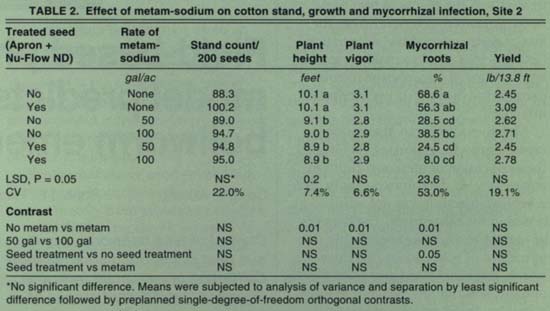
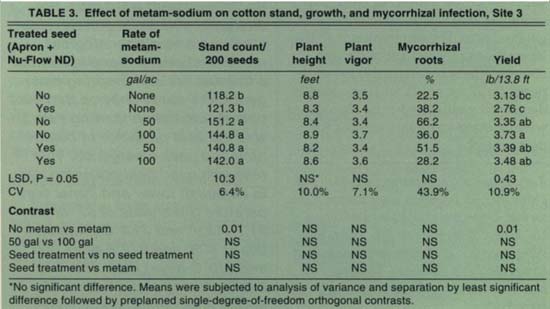
Both the seed treatment and the metam-sodium soil treatment significantly increased plant stands (healthy, germinated seeds) at Site 1 (table 1). However, metam-sodium significantly reduced plant height and plant vigor. Yields were also reduced in plots treated with metam-sodium at Site 1. Mycorrhizal infection of the roots was significantly reduced by metam-sodium (81.7% to 22.0%). At Site 2, stands were not affected by the seed treatment or metam-sodium (table 2). Plant height and vigor, however, were adversely affected by metam-sodium. The percentage of roots infected by mycorrhizal fungi was also reduced by metam-sodium (62.5% to 24.9%). At Site 3, stands were significantly improved with application of metam-sodium but not the seed treatment (table 3). Neither treatment affected plant growth, but yields were significantly increased by metam-sodium. Unlike the other two locations, mycorrhizae development was not affected by the metam-sodium treatment, possibly due to high soil populations of mycorrhizal fungi. There were no significant differences between application rates of metam-sodium in any parameter at any site. The variable results from the metam-sodium treatment were not related to soil type, although it has been observed that the efficacy of metam-sodium is enhanced in soils in good tilth.
While metam-sodium consistently provides good weed control in the seedrow in cotton fields in the San Joaquin Valley, there are additional positive and negative effects of metam-sodium use on cotton culture. In two of these trials, metam-sodium increased cotton stands (presumably by reducing seedling disease) compared to the nontreated seed. In one of these trials it was more effective than the standard fungicide seed dressing. However, the application of metam-sodium also negatively affected plant development in two of the trials. In one trial yields were also reduced.
These negative effects were consistently associated with reduced mycorrhizae formation. Cotton is apparently very dependent on mycorrhizal development, which benefits the plant by increasing the uptake of certain minerals, especially phosphorus. These fungi are a natural component of all agricultural soils in the San Joaquin Valley, and normally infect the roots of most crops, including cotton. As mycorrhizal fungi grow within and outside the root system, they increase the effective absorptive surface area of the roots many times. The reduction in the intensity of mycorrhizal infection in these experiments by metam-sodium resulted in shorter plants, fewer nodes and less yield. Although roots of the cotton plants grew outside the relatively small zone of metam-sodium treated soil, the plants in two trials never appeared to recover from the loss of early mycorrhizal association. Growers may need to make preplant applications of phosphorus to compensate for decreased mycorrhizal infection.
Metam-sodium can also cause increased growth responses. At Site 3, the application of metam-sodium increased yields. Not unexpectedly, mycorrhizal infection was not reduced as it was in the other trials. The benefits of metam-sodium were probably due to control of root pathogens, since stands were increased and plant development was not affected. For disease suppression, 50 gallons of metam-sodium per acre was as effective as 100 gallons/acre. The economics of all the interactions of weed and pathogen control, effects on mycorrhizae, additional fertilizer needs and increased or decreased yields need to be considered when metam-sodium is applied for pest control.