All Issues
Spring insecticide treatments control adelgids on Douglas fir
Publication Information
California Agriculture 50(5):34-37. https://doi.org/10.3733/ca.v050n05p34
Published September 01, 1996
Abstract
Insecticide applications made to Douglas fir Christmas trees vary in their ability to suppress Cooley spruce gall adeigid, a sucking insect that causes needle distortion and discoloration. A field experiment in Napa County showed that organophosphate systemics are ineffective at controlling adelgids, but imidacloprid, a newly registered systemic insecticide, is highly effective. Horticultural oil is effective, but causes slight needle yellowing. This side effect can be mitigated by precisely timing the oil application before bud break when the overwintering adelgids are transforming to adults but before they lay eggs.
Full text
Left, needle distortions caused by the Cooley spruce gall adelgid feeding on Douglas fir and white waxy material left by females. Above, yellow spots on the upper side of the needles are caused by adelgids feeding on the underside.
The Cooley spruce gall adeigid, Adelges cooleyi (Gillette), can be a serious sucking-insect pest on Douglas firs grown in Christmas tree farms and in forest tree nurseries. High populations can cause severe needle distortion and chlorotic spots rendering the trees unmarketable. There is little information on the best method and timing for the control of this pest under California conditions. Presently many labeled insecticides are registered for aphid control, but there is a lack of comparative data on their efficacy in controlling Cooley spruce gall adeigid. This study was undertaken to compare registered materials, including a new systemic insecticide, and to explore the use of horticultural oil as an inexpensive and less toxic alternative to conventional synthetic insecticides.
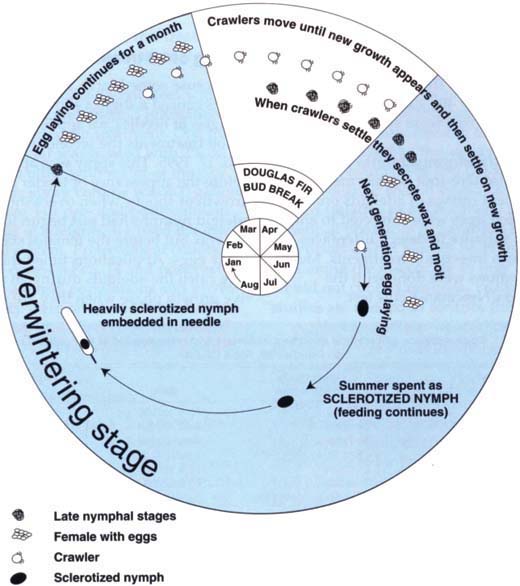
Adeigid life cycle
The Cooley spruce gall adeigid usually alternates generations between spruce (the primary host on which these insects form galls and then undergo sexual reproduction) and Douglas fir (the alternate host on which they reproduce asexually). Alternation of generations between these two hosts is not essential for insect survival or population increase. In Napa County, we have only observed the asexual cycle in Douglas fir Christmas tree farms.
Adeigid nymphs overwinter on the current year's growth of Douglas fir needles (fig. 1). The overwintering nymphs are dark brown in color and are heavily sclerotized (the hardening of their exoskeletons). In mid-February the nymphs start secreting a waxy fringe. Nymphs molt in approximately 1 week and become adults. They then begin to lay eggs under heavy waxlike strands. Egg laying continues for approximately 1 month. In our observations, the average number of eggs laid was 44. Eggs begin hatching 2 to 3 weeks after being laid. One percent egg hatch was noted in early March, and 95% egg hatch was recorded by mid-April.
Newly hatched (first stage) nymphs are light brown with a darker head. They are called crawlers because they move until they find a suitable place to settle. The majority of crawlers do not settle until the new foliage emerges. This new growth becomes available to crawlers at different times on individual Douglas fir trees, which are seed propagated and are genetically variable. Douglas fir in Napa during 1994 and 1995 began spring growth over a period of about 5 weeks. Earliest terminal bud break began during the third week in March; the latest bud break occurred in the last week in April.
Once crawlers move to new growth they gradually darken and secrete a white waxy fringe around the edge of the body. They become flattened, with the legs folded under the body, and fix themselves firmly to the leaf surface. The second-stage nymphs are light brown and the body is covered with a waxlike secretion. Nymphs soon change to egg-laying adults covered with curled waxy strands. Adults were seen about 10 days after crawlers settled on the new growth. It is this spring-generation feeding by the nymphs and adults on the newly expanded needles of Douglas fir that causes the greatest damage. High populations cause needles to become distorted and discolored; yellow spots appear at sites where insects have fed.
Summer-generation adults begin laying eggs in early-to-mid-May and produce only a quarter the number of eggs (average 12 per female) that the first generation produces. Crawlers appear 2 to 3 weeks after egg laying is observed (late May to early June). Because the needles harden as summer progresses, the development of the second generation is prolonged and there is high nymphal mortality. Adelgids remain in heavily sclerotized immature stages through the late summer, fall and winter. Nymphs are more prevalent on the north side of the tree. Nymphs that survive the summer are generally found at the base of the needle or below the tuft of waxy strands left by the female. Feeding continues throughout the summer, as evidenced by a droplet of honey-dew secretion at the rear of the nymph's abdomen. By late October the nymphs are heavily sclerotized, no secretions are visible and the nymphs are firmly embedded in the needle on either the upper or lower side of the base of the needle. When on the upper side of the needle, they embed themselves on the middle vein.
Systemics and oil combinations
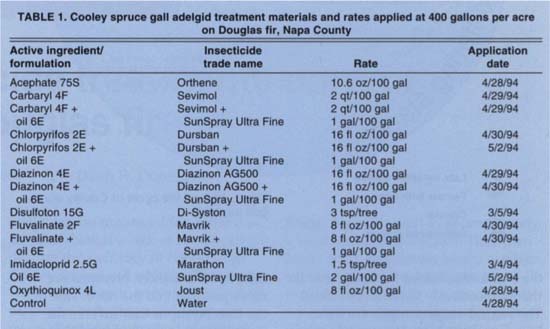
A field experiment was conducted with 2 acres of Douglas fir trees planted in 5-feet-by-5-feet spacing on Haire loam soil located near Napa. We divided the field into 4 blocks and each block into 14 insecticide treatment plots (table 1) according to a randomized complete block design. Each plot had approximately 50 trees, 3 to 10 feet tall. The entire plot was treated and 4 trees (5 to 8 feet tall) per plot with high adelgid populations were selected for season-long monitoring.
Adelgid crawler (left) and settling nymph (right) with a droplet of honeydew secretion at the rear of its abdomen.
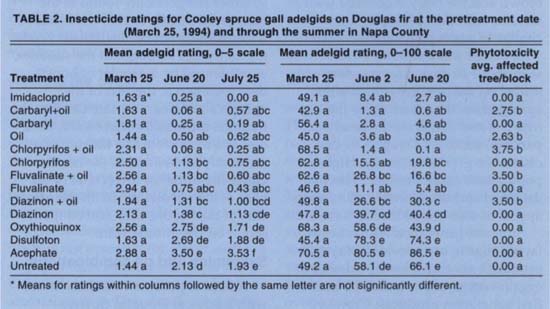
Adelgid control was evaluated using two systems: insect count rating and visual rating. Both systems were used on March 25 and June 20 to determine whether their results were consistent. The count rating system was designed to provide more information than a presence-absence rating system, and generated data amenable to analysis using methods designed for other 0-to-100 scales, such as percentages.
For the insect count rating, we examined the first 7 inches of the tips of 10 branches from each sample tree for the presence of adelgids. Adelgid numbers were rated by assigning each shoot a score of 0, 1 or 10. No adelgids present on a shoot scored 0, one adelgid present was assigned a score of 1 and shoots with two or more adelgids were assigned a score of 10. The jump in rating from 1 to 10 reflects the typical presence of aggregations of adelgids when treatments were ineffective, and provides a convenient 0-to-100 point rating for each tree when the 10 shoot scores were totaled. Insect count ratings were done on Feb. 24 (pretreatment count), March 25, June 2 and June 20.
For the visual rating, each sample tree was rated from 0 to 5. A minimum score of 0 was assigned when no adelgid was visible when the canopy was opened three times. Opening the canopy involved spreading the branches to inspect the interior of the canopy. A score of 1 was assigned when adelgids were visible within the canopy but not on the outside. Scores of 2, 3 and 4 corresponded to less than 15%, 45% and 75% of the branches infested with adelgids, respectively. A maximum score of 5 was assigned when adelgids were visible on more than 75% of the canopy. The visual ratings were done on March 25, June 20 and July 25.
Phytotoxicity was rated on the sample trees on July 25. Since discoloration was uniform, those trees with no discoloration were rated as 0; those with yellow-green discoloration were rated as 1.
Granular pesticide treatments were applied first to allow time for systemic uptake. The two granular treatments, imidacloprid (0.4 lb ai/ac) and disulfoton (6.5 lb ai/ac), were applied March 4 and 5 in a grid of 2-inch-deep holes, 2 1/2 feet apart. To activate the insecticide, the field was irrigated the day after treatment. One row of trees on all four sides of the plot was left untreated as a buffer. Sample trees in all plots were at least two trees away from the buffer zone.
Foliar spray treatments were applied between April 28 and May 2, timed at maximum egg hatch and before wax formation was observed on newly hatched adelgids. To estimate egg hatch, we took samples twice a week. Sprays were applied with a hand-held nozzle to run-off, using approximately 400 gallons of water per acre. Controls were sprayed with water.
Nontransformed ratings on March 25 and square-root transformed post-treatment ratings of adelgids on sampled trees were subjected to analysis of variance to detect differences between insecticide treatments. Mean separations were done with the Student-Newman-Keuls test.
TABLE 1. Cooley spruce gall adelgid treatment materials and rates applied at 400 gallons per acre on Douglas fir, Napa County
TABLE 2. Insecticide ratings for Cooley spruce gall adelgids on Douglas fir at the pretreatment date (March 25, 1934) and through the summer in Napa County
TABLE 3. Ratings for Cooley spruce gall adelgids treated with SunSpray Ultra Fine oil on Douglas fir at the pretreatment date (Feb. 14, 1995) and through the spring In Napa County
Timing of oil treatment
Because some March 1994 treatments caused a slight yellowing of Douglas fir needles, we applied follow-up oil treatments the second year on Feb. 17, 1995. This spray was applied before the appearance of tender new growth of shoots, when overwintering adelgid nymphs had just begun forming wax but before the females started to lay eggs. Applications timed this way catch the adelgids during an active growth phase when they are most sensitive to pesticides; this treatment opportunity window lasts only about 7 days.
Eight paired adjacent trees with similar size and adelgid infestations were selected for treatment. One tree of each pair was treated with 2% oil and the other with water as the control; the spray volume was 400 gallons per acre. For pre- and posttreatment ratings of adelgids, we used the insect count system. Pretreatment counts were done on Feb. 14 and posttreatment adelgid populations and phyto-toxicity were rated on March 4 and April 18. The effect of oil on adelgid mortality was analyzed with a paired t-test.
Vried effectiveness
As shown in table 2, insecticides varied in their ability to suppress Cooley spruce gall adelgids. The two rating systems were highly correlated (r2 = 0.78 with one outlier, March 25 data; r2 = 0.93, June 20 data), which resulted in consistent measures of pest populations, including similar ranking and statistical separation of treatment means.
Except for disulfoton and acephate, two systemic organophosphate insecticides, all treatments demonstrated some degree of suppression of Cooley spruce gall adelgids.
Effective control of Cooley spruce gall adelgid was obtained with the following treatments: imidacloprid, car-baryl + oil, carbaryl, chlorpyrifos + oil, fluvalinate, fluvalinate + oil, chlorpyrifos and oil. Oil enhanced the activity of carbaryl and chlorpyrifos, providing excellent early-to-midseason control of adelgids.
Imidacloprid, an insecticide recently registered in California, has unusual properties that may make it suitable for use in Christmas tree plantations. Although the present label only allows foliar applications to Christmas trees, on most crops it has been most effective when applied to soil for uptake through the roots. When protected from the sun, it degrades slowly so an application results in extended uptake throughout the growing season. For example, the higher rates labeled for ornamental landscape trees commonly show insecticidal effects lasting into the year following application (data not shown). The soil application of imidacloprid granules at agricultural use rates (0.4 lb ai/ac) resulted in continued movement into test trees through the summer, as indicated by the steady decline in adelgid counts. The low dosage and soil application should minimize the hazard (relative to foliar sprays) to beneficial predators or parasites. Most beneficials do not feed on plant tissue containing the insecticide so they would not contact residues. Further studies are needed to confirm this.
Application below the soil surface in this trial was accomplished with granular formulations by placing a measured dose in a planting bar hole between each two trees within the row. Sprayable formulations, such as the flowable imidacloprid product now registered for use in Christmas tree plantations, could be applied efficiently through precision soil injection devices. These techniques lead to minimal exposure of applicators and wildlife to insecticides, and should be investigated in further trials. Subsurface application does not require expensive equipment, nor does it require smooth ground suitable for driving tractors or sprayers. This is a distinct advantage, considering that many Christmas tree plantations are small farms or are located on rough terrain.
Carbaryl, a persistent carbamate insecticide, often induces secondary outbreaks of spider mites by eliminating mite predators. The spruce spider mite, Oligonychus ununguis, is a particularly damaging species to Douglas fir. Therefore applications of carbaryl without oil might require additional miticide sprays later in the season.
Chlorpyrifos or fluvalinate applied without oil can be considered reasonable choices for use in Douglas fir Christmas tree plantations. Both materials show some efficacy against both adelgids and spider mites. However, pyrethroids such as fluvalinate tend to select for resistant mite populations very rapidly, so rotation later in the season with other classes of miticides (if needed) should be considered.
Treatments found unacceptable based on the poor level of control were, in order of increasing efficacy: oxythioquinox, diazinon and diazinon + oil. The poor performance of the diazinon + oil combination is paradoxical, since horticultural oil by itself was one of the more effective treatments.
Oil by itself or in combination with any insecticide was phytotoxic to Douglas fir in the 1994 trial (table 2). The symptom of damage was uniform discoloration of needles throughout the tree, changing from the normal blue-green to yellow-green. This slight discoloration remained for the entire season. Needle discoloration was not visible in new growth the following year.
The 1995 oil treatment timed to avoid the new growth gave moderate adelgid control (table 3). However, older growth needles turned yellow-green within 48 hours after the oil application in all the treated trees. Once the new growth covered it, needle discoloration was no longer noticeable and appeared to affect neither tree growth nor needle shedding.
Effective treatment options
Effective treatments that did not cause phytotoxicity were (ranked in order of decreasing efficacy): imidacloprid, carbaryl, chlorpyrifos and fluvalinate. Horticultural oil suppressed adelgids but also caused Douglas fir needle discoloration. If oil is applied after bud break, the discoloration is visible at harvest. If oil is applied in the very narrow time frame before bud break, when the overwintering adelgids are transforming to adults but before they lay eggs, needle discoloration occurs but is later masked by new growth. In this case discoloration is not visible at harvest.