All Issues
Soil fumigants provide multiple benefits; alternatives give mixed results
Publication Information
California Agriculture 48(3):22-28.
Published May 01, 1994
PDF | Citation | Permissions
Abstract
Since the 1950s growers have routinely used soil fumigants such as methyl bromide (MBr) and 1,3-dichloropropene (1,3-D) before replanting orchards and vineyards. Fumigants double plant growth in the early years after replanting and provide several years of nematode relief when resistant rootstocks are unavailable. However, the recent suspension of 1,3-D and the mandated phase-out of methyl bromide by 2001 have clouded the future of fumigant use. To develop alternatives, we must first document the pest control value and plant growth benefit of fumigation. Over the last decade, we have initiated several 2-year field trials in replant sites in the San Joaquin Valley. Our results quantify fumigation benefits and point to the feasibility of some alternatives, including a portable soil drencher.
Full text
Northern California black walnut rootings planted in nonfumigated soil, on the right, show signs of replant problem compared to those on the left grown in fumigated soil.
Scientists cannot entirely explain the powerful benefit of fumigating soil before replanting perennial crops. The doubling of plant growth in the years that follow is only partly accounted for by the elimination of plant pathogens, nematodes and insect pests. Evidence suggests that myriad detrimental factors commonly present in old orchard sites are remedied by fumigation and soil profile disruption.
However, the future of this widely used technology is in doubt. In 1990, California use of 1,3-D was suspended due to its occurrence in off-site air samples. In addition, MBr use in the United States will be phased out by the year 2001 under provisions of the 1990 Clean Air Act.
At present, there are few satisfactory alternatives to the long-standing practice of soil fumigation. Fumigants are capable of destroying most life stages of soil-dwelling organisms as well as the roots of old grapevines which can otherwise remain alive in soil for 10 years after the vine trunk has been ripped out of the ground. Roots of peach, plum, apple and walnut deteriorate faster than those of grape, but viable roots are likely to be present for at least 3 years after tree removal. These woody roots provide a food source for a variety of soilborne plant pathogens and insects. Viruses, bacteria or actinomycetes (rod-shaped, branching bacteria which may be obligatory plant parasites or simply grow in association with the roots) will also persist in soil as long as the old roots remain viable. Fumigation at 250 to 700 lb/ac MBr or 1,3-D to properly prepared soil addresses these potential problems. It kills old roots in the surface 4 to 6 feet of soil profile, and most life stages of soil-borne pathogens and insects, promoting growth and vigor in a replanted orchard.
However, if an orchard is replanted in nonfumigated soil, the result is quite different. This is due to a phenomenon known as the “replant problem,” which occurs to varying degrees worldwide. The replant problem is illustrated when soil is collected from anywhere along an old orchard or vineyard floor and placed in a pot in the greenhouse: Any woody perennial replanted into it will not grow as well as if it were planted to soil that had never been in an orchard or vineyard. Two of the perennials that present the worst replant problems (when followed with any other perennial) are peach and walnut. If a replanted perennial is the same type as the previous one (e.g., grape to grape), the plant growth reductions can be striking. This latter phenomenon is referred to as a “specific replant problem.”
T his tractor-mounted device, which picks up and reels in drip tubing, is an example of a portable soil drencher.
There are biological, chemical and physical components of the replant problem and they are not necessarily the same from site to site or region to region. For example, replanting peach to peach in the Sacramento Valley is not as perilous as doing so in the San Joaquin Valley. In some replant sites, all that is needed to correct the problem is deep soil ripping or backhoeing of the planting sites.
The symptoms of a replant problem include poor growth with nutritional deficiencies such as phosphorus and zinc distributed non-uniformly across the field. Nematodes, phytophthora, phylloxera and other soil pests may be abundant along the roots. Plants do not develop adequate root systems and all of this can be visible by midsummer of the first year. In some situations, these problems will disappear after one or two years. In other cases, especially where a known soil pathogen is present, the plants may become an economic liability to the grower unless a rootstock with resistance to the dominant pest was planted.
Delivery of biocides via this portable soil drenching device is among alternatives to soil fumigation that researchers are evaluating.
The combination of soil profile disruption and proper soil fumigation at the planting sites will stop the general replant problem and greatly minimize the specific replant problem. Soil fumigation will provide 6 months to 6 years of nematode relief, depending on the quality of the treatment. If nematodes have returned to the site within 6 months after planting a susceptible rootstock into a soil conducive to their development, the plants may grow well for the first 2 years and then quickly or gradually decline in vigor. In fumigated and nonfumigated blocks grown side by side, a grower may continue to see the growth difference decades later. The grower can try a number of post-plant treatments to revitalize the poorer growing block, but usually with only mediocre success. For purposes of comparison, 4 years of dry fallowing prior to replanting is generally adequate to avoid most of the replant problem.
In this paper, numerous pre-plant treatments will be compared for their long-term nematode control value and the vitality they impart to replanted trees and vines. These studies are the results of cooperative field trials located at sites where human and vehicle traffic could be kept to a minimum to avoid contamination from plot to plot. MBr and 1,3-D are examples of “true” soil fumigants. This means that their movement through soil is primarily as a gas. These two biocides are released into soil behind tractor-pulled shanks. The biocides become gaseous and move through soil air spaces (fuming), while dissolving into associated films of soil water. Within the soil water film, soil microbes are contacted and killed.
Although it is a very good biocide, the fumigant metam sodium (MIT) actually has poor fuming action. It permeates the soil only if it is moved with water. Several nematicides, including fenamiphos, oxamyl and ethoprop, are not biocides or fumigants but rather nematicides moved best with water. Carbon bisulfide has excellent fuming action, but can be ignited by a spark. However, the formulated product Enzone has limited fuming action, is nonexplosive, and also is moved best through soil by adding it to water.
Marigold (Tagetes spp.), which is antagonistic to various nematode species, may be grown as a rotation crop or refuse of its above-ground parts may be added by incorporation as a soil amendment. Extracts of marigold can also be drenched onto soil. Marigold provides an example of a potential nonconventional alternative to soil fumigation.
Soil drenching is a procedure for mixing biocides into irrigation water and delivering the water onto the field surface through basins, sprinklers or large tanks on wheels. A new procedure for drenching involves the use of a portable soil drenching device (PSDD) ([[see photos 2 & 3]]). These devices can deliver a wider variety of biocidal agents with greater uniformity while minimizing fumigant escape (off-gassing); minimal off-gassing was recorded in preliminary measurements by air monitoring instruments. Using the PSDD, a dripper emitter is temporarily located on each square foot of field surface that is drenched (see photos, p. 23 ). The drencher mixes biocide with water, and distributes the mixture through the soil profile over an 8-hour period. The cost of using the PSDD is estimated at $150 per treated acre plus chemicals, which is similar to that of nontarped methyl bromide applications, the prevalent practice of the tree and vine industry at present.
Materials and methods
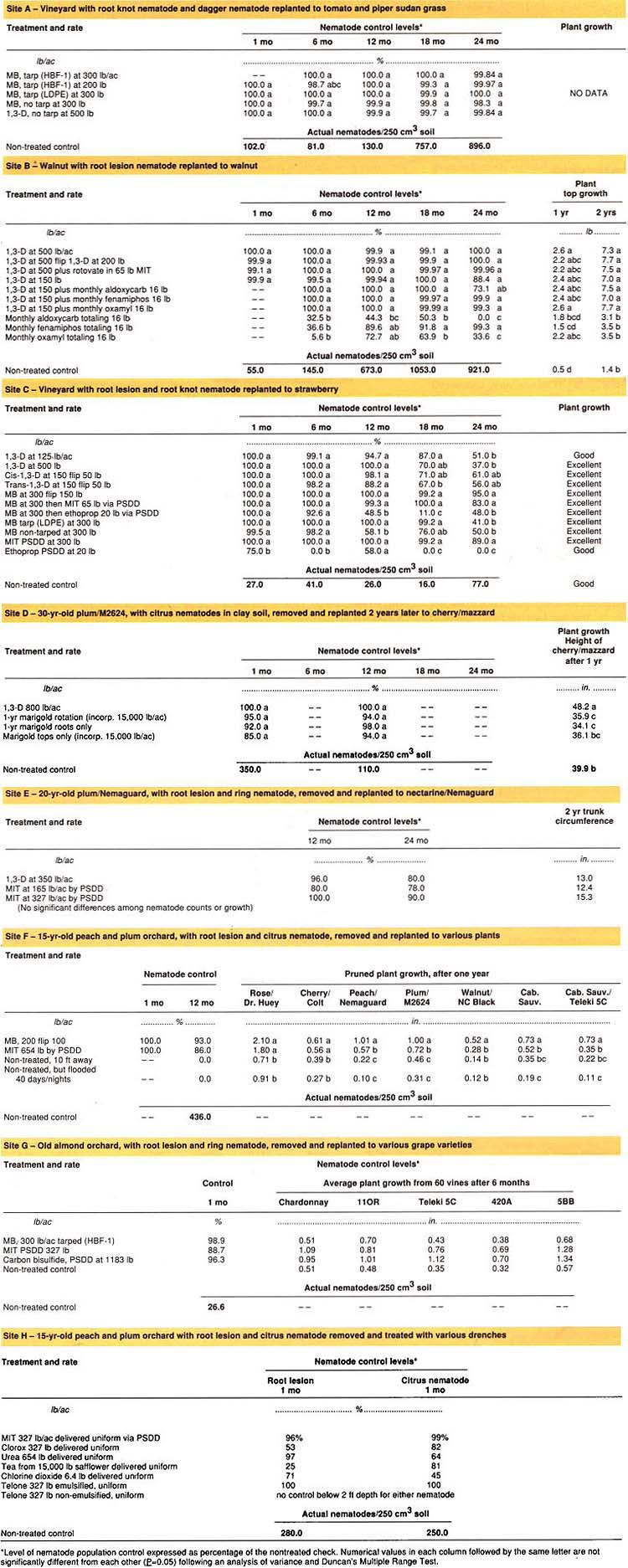
Studies were conducted at eight locations, which are referred to here as Sites A through H (see table 1). All the locations were in Fresno County, except Site G, which was in Stanislaus County. Sites A, B, C, F and H were near Parlier. Site D was close to Orange Cove, Site E was near Reedley and Site G was close to Ceres.
The overall goal of this research was to establish the benefit of current soil fumigation and investigate the feasibility of alternatives. The specific goal in each trial was to reduce grower costs without losing long-term benefit. Evaluations were conducted on more effective plastic tarpaulin, lower treatment rates, dripper applications with nematicides instead of biocides, natural nematicides, and eventually a number of biocides and nematicides applied through the portable soil drencher.
During the first month or two after each treatment, and before replanting, soil samples were collected at 1-foot increments down to a 5-foot soil depth. These five soil samples were analyzed separately, but to simplify reporting the results have been averaged together and listed under the 1-month sampling column. Once the treated sites were replanted, all subsequent soil samples were collected from the 0- to 2-foot depth of each treated replicate.
Collected soil was extracted for nematodes using a combination Cobb Sieving and 5-day mist extraction. Collected nematodes were placed beneath a microscope for speciation and counting. Nematode counts for the nontreated control are indicated as nematodes/250cm3 soil. All other data for that site are depicted as percent control across the four to eight replicates for that treatment when compared to the nontreated. Counts for Paratrichodorus minor (Stubby Root Nematode) and Paratylenchus hamatus (Pin Nematode) were excluded from all data sets.
Plant growth measurements were taken 1 or 2 years after treatment and included plant weight, pruning weight, plant height and trunk circumference.
The soil in Site D was a Porterville clay. All other sites involved a Hanford sandy loam soil. The treatments were as follows.
Site A
The Site A study was designed to determine the value of a new high barrier, greater density tarp (HBF-1) compared to the lower density tarp, which was in common use until the late 1980s.
TABLE 1. Data on plant growth and nematode return in the 24 months following various soil treatments
Site B
The Site B study was conducted to determine if 1,3-D fumigations at low rates (150 lb/ac) plus monthly 1 lb/ac additions of conventional nematicides through an irrigation dripper could be as effective as a conventional 250- to 700-lb-rate preplant fumigation with 1,3-D. In one of the 10 treatments, the 1,3-D was followed in a week by a 6-inch-deep rotovater with MIT (metam sodium) sprayed just before the front tines for incorporation. (The rotovater has moving tines which mix up soil to a depth of several inches.)
Site C
At Site C, dual treatments with MBr and 1,3-D were compared to conventional treatments. Dual treatments involve a normal treatment, followed two weeks later by a plowing designed to flip the surface 1 foot of soil completely upside-down. The plowing is followed within a day by a second treatment of usually the same fumigant but at a reduced treatment rate. The first comparisons with a Portable Soil Drenching Device (PSDD) began in Site C. It was used to deliver 3 inches of water with various biocides or nematicides to the surface of dry soil after a deep fumigation or 7 inches of water with various biocides to a moist soil. The PSDD delivered the water at the surface with the biocide uniformly metered over a 3- or 9-hour period. When using a PSDD, dripper emitters are placed on each square foot of field surface.
Site D
Site D involved a comparison of 1,3-D with marigold, Tagetes tenuifolia c.v. Nemakill as a rotation crop or as a refuse amendment. The fresh weight of the aboveground marigold plant was approximately 15,000 lb/ac as it was incorporated and irrigated in four months before planting of cherry/mazzard.
Sites E through H
At Sites E through H, we performed field tests of PSDD using various biocides.
Site F
In Site F, five each of seven different host crops were grown in each plot after the treatments. MBr was applied at 200 lb/ac at a depth of 3 feet, the soil flipped in 7 days and retreated at 100 lb/ac at the 18-inch depth. One set of plants was planted 10 feet away from the old tree rows while all other plants were within 2 feet of the old planted row. At this site, the untreated block was flooded for 40 days and nights with 16 feet of water in an unsuccessful attempt to kill old roots. These flooded treatments can be considered a nontreated control.
Site G
At Site G young rooted grapevines were planted 1 month after mid-summer treatments including a relatively high rate of carbon bisulfide (Enzone). Vine top weights from 60 vines in each replicate were collected and weighed.
Site H
In Site H a variety of natural or nonconventional biocidal agents were applied via PSDD after killing the old orchard trees with a 2% foliar application of glyphosate. A “uniform delivery” was achieved by mixing the biocide into every drop of water delivered except for the last 30 minutes of delivery. The drenched 1,3-D included a non-emulsified and an emulsified product.
Sites F, G and H are still under investigation at the time of this writing.
Results and discussion
Field replant sites receiving 250 to 700 lb/ac of properly applied 1,3-D or MBr frequently exhibited nematode population reductions of 95% up to 2 years after fumigation (see table 1). These nematode reductions are accompanied by plant growth benefit. Although the use of marigold as a rotation crop or as a refuse amendment in the year before planting cherry resulted in reduced nematode populations, it significantly reduced plant growth when compared to fallowing for 1 year or the pre-plant use of 1,3-D (site D).
Soil drenching, when used to apply MIT (the active ingredient released by metam sodium in soil) with 6 inches of water to properly prepared soil, came very close to being as effective as soil fumigation (Sites C, E, F and G). However, the MIT treatments do not kill old tree roots below 2 feet soil depth when applied at 327 lb/ac (100 gal/ac Vapam). At double the treatment rate (654 lb/ac), old roots may be killed down to 4 feet; however, replants placed in the soil 6 months later (Site F) grew poorer than those planted to MBr fumigated soil. At the 327 lb/ac rate grapevines planted one month after treatment (Site F) or Nemaguard rootstock planted 3 months after treatment (Site E) did grow as well as the fumigated comparisons (no significant differences).
Compared to the untreated control, replants of black walnut were doubled in size 2 years after a 1,3-D fumigation at conventional rates (Site B). Treatment rates of 150 lb/ac of 1,3-D only gave nematode control in the surface 3 feet of soil profile and did not kill all the old roots in that zone. Knowing this, dripper applied treatments of conventional nematicides at 1 lb/ac were applied monthly to repel intrusion of the root lesion nematode. The dripper treatments were effective in the narrow zone receiving treatment, but not in sites more than 2 feet away from the drip emitter (data not shown). Fenamiphos (Nemacur), when applied in this manner to 2-year-old nursery stock of walnut, reduced plant growth during the first year of treatments but improved walnut growth in the second year (Site B). In Site F, walnut trees planted into MBr-fumigated soil that had not previously been planted to walnut were four-fold larger than the nontreated. Compared to MBr treatments the growth of walnut trees in soil treated with 654 lb MIT/ac (200 gallons/ac Vapam) was reduced although nematode control was excellent. In Site F, the only plants infected with root lesion 1 year after the MIT drench or MBr fumigation were those adjacent to one edge of the field where a buried concrete pipe line existed.
Nemaguard Peach, NC Black Walnut and Teleki 5C grape were the three rootstocks that showed greatest growth benefit from a pre-plant soil fumigation. These are examples of plants that appear to be more sensitive to the replant problem in general. All three have resistance to root knot nematode, but are susceptible to root lesion nematode. In Site F, high rates of MIT reduced growth of the subsequent planting, but it should also be noted that young grape rootings do not always grow well after MBr (see Site G), whereas they do grow well after 1,3-D fumigation (not shown). In none of these trials did we make an attempt to correct nutritional deficiencies with fertilization, since such deficiencies are usually a result of the killing of beneficial soil microorganisms such as mycorrhizae. MIT applied by PSDD at 327 lb/ac gave control of nutgrass Cyperus esculentus similar to that achieved with tarped MBr. The microbial-rich surface soil receives high dosage levels during the drenching process (especially at 654 lb/ac rate of MIT) and we should anticipate kill of some beneficial organisms at high treatment rates. The relatively high rate of carbon bisulfide used in Site G resulted in visible growth improvement within that treatment compared to MBr treated soil. The results are not statistically significant probably because the young vines were only in the ground for two summer months before being weighed.
The lack of root killing power in MIT at the 327 lb/ac rate may be more of a problem for old orchard sites than for vineyards where smaller roots are more common. It should be noted that trees and vines irrigated by dripper or mini-sprinkler have a greater number of small-sized roots which would succumb to MIT at a lower dosage rate. As recipes and equipment for PSDD have gradually evolved the potential for drenching with existing low volume irrigation systems has also become apparent. After the last harvest, existing irrigation systems can become the vehicle for drenching with MIT and other biocides.
Drenching will become one of the methods used to replace soil fumigation. One primary reason is that a fumigant's ability to move well through soil as a gaseous biocide also causes it to appear off-target in ambient air. Unlike fumigations, drenching locks the biocide into soil. If the biocide has been properly selected (e.g. short half-life) it will degrade in place. Drenching also permits the use of less-volatile biocides. In using the PSDD to deliver MIT, we have repeatedly observed a reduction in the quantity of biocide volatilized from treatment sites. The odors of Vapam are not detected 100 ft from the treated area. Our early studies indicated that whether soils were 40°F or 80°F, the efficacy of the MIT was adequate (data not shown) against nematodes in the surface 4.5 feet of soil. Soils must be in proper condition to receive the drenching. If water will not move through the soil with uniformity, then neither will a biocide having low volatility. In regard to proper soil conditions, there is much less flexibility with MIT than with MBr.
The timing of 1,3-D and MBr treatments has been consistently recommended for the fall months before 2 inches of rainfall. Fumigation has usually been the last cultural event before replanting. Drench treatments have traditionally followed a similar recipe; however, evidence is accumulating to indicate that there may be a waiting period and/or a rotation crop needed between drenching and replanting. Drenches with MIT may be more appropriately applied before or just after removal of the old orchard or vineyard. After plant removal and soil profile modification, annual crops that are deep rooting and nonhost to indigenous soil pests may need to be grown for six months or more before replanting of trees and vines. The waiting period would further reduce populations of endoparasitic nematodes, restore beneficial soil microbes and restore soil structure prior to replanting. It will require several years to field-evaluate this change in general procedures.
Although average treatment rates of 400 lb/ac of biocidal agents seem relatively high, the “nonchemical” alternative we have studied most is 15,000 lb/ac of aboveground fresh weight of marigold. A simple water extract of marigold is nematicidal at 10g fresh weight/liter of water. Equivalent nematicidal activity can be attained with MBr or 1,3-D at 10 mg/l. Whether we apply the marigold as an extract within a drench or incorporate and add water to the soil, the marigold contains numerous biocidal ingredients that appear to persist in soil unless about 40 inches of rain falls between incorporation and replanting. These biocidal agents do not kill old roots, but they inhibit growth of replanted trees and vines. We are continuing to study marigold drenches followed by 40-inch irrigations, but it is incorrect to assume that because ingredients occur naturally they are safe for the environment.
The soil fumigants have offered growers flexibility and have been relatively easy to apply. There are no examples of MBr or 1,3-D as contaminants of groundwater because both fumigants also degrade in soil. Both products are, however, relatively volatile and apparently difficult to maintain below ground once they are applied. Procedures for keeping them below the soil surface include reductions in treatment rates. As treatment rates are reduced, the spectrum of soil-borne pests they control will also be reduced.
Conclusions
For a variety of reasons, soil fumigation has proven to be an economically viable pre-plant treatment. Even “resistant” rootstocks grow poorly their first year or two without such soil treatments. Crop rotations of 1 or 2 years are relatively expensive and, as with marigold in an arid climate, may not yield positive growth benefits. Our more deliberate approach to soil drenching has provided a broader list of potential biocides and treatment combinations, while also indicating how the more volatile biocides may need to be applied to minimize off-gassing. Inches beneath our feet right here on Earth there are microbiological systems and interactions that we know absolutely nothing about. Until this deficiency is corrected, the finding of alternatives to fumigation will be like shooting in the dark.