All Issues
Biological and chemical pruning wound protectants reduce infection of grapevine trunk disease pathogens
Publication Information
California Agriculture 75(3):128-134. https://doi.org/10.3733/ca.2021a0018
Published online January 07, 2022
NALT Keywords
Abstract
Grapevine trunk diseases (GTDs) are currently considered some of the most important challenges for viticulture, curtailing vineyard longevity and productivity in nearly every raisin, table and wine grape production region in California and worldwide. Pruning wounds provide the main entry point for fungal pathogens responsible for these diseases; pathogens enter the wounds following precipitation events. The aim of this study was to evaluate the efficacy of selected chemical and experimental biological fungicides for protection of pruning wounds against two of the most common and virulent fungal pathogens causing GTDs: Eutypa lata and Neofusicoccum parvum. This study was conducted on sauvignon blanc at the UC Davis Department of Plant Pathology Field Station. Results showed that several chemical and biological fungicides, notably the chemical fungicide Luna Sensation, the biofungicide Vintec and a combination of the biofungicides Bio-Tam and CrabLife Powder, provided significant protection against at least one of the two canker pathogens used in this study. However, the majority of products tested did not provide simultaneous control of both E. lata and N. parvum pathogens, highlighting the continuing challenge of controlling GTDs.
Full text
Cultivated for over 7,000 years, grapevines are today one of the most extensively grown and economically important woody fruit crops in the world. In 2018, approximately 18.3 million acres (7.4 million hectares) of grapevines were cultivated and 77.8 million tons of fruit were harvested around the globe (OIV 2019). In the United States, California leads the way in grape production; in 2019, grapevine cultivation totaled 918,000 acres (37,000 hectares) (CDFA and USDA NASS 2019). Vitis vinifera (common grapevine) cultivars are the most widely planted in California and elsewhere, yielding a high commercial value for fresh table grape, dried fruit and wine production (Gramaje et al. 2018; Mullins et al. 1992).
Results from a UC Davis field trial of sauvignon blanc indicate that Trichoderma based biological fungicides can protect against both E. lata and N. parvum, but the majority of treatments tested were not effective in controlling both pathogens simultaneously. Photo: Robert Blundell.
However, V. vinifera is susceptible to infection by a large variety of pathogenic microorganisms, including fungi, oomycetes, viruses and bacteria (Gaulon-Brain et al. 2007). Among the most destructive infections are the grapevine trunk diseases (GTDs). Caused by a broad range of taxonomically unrelated fungi, GTDs present a major threat to the economic sustainability of table and wine grape productions worldwide (Trouillas et al. 2010). They cause serious economic losses due to a significant reduction of both yields and quality of grapes; they also increase crop management costs for cultural and chemical preventative measures (Bertsch et al. 2013; Gubler et al. 2005; Kaplan et al. 2016; Siebert 2001). Important GTDs include Eutypa dieback, Botryosphaeria dieback, Esca, and Phomopsis cane and leaf spot. In this study, we tested two of the most prevalent GTD fungal pathogens found in California crops, Eutypa lata and Neofusicoccum parvum, which are causal agents of Eutypa dieback and Botryosphaeria dieback, respectively (Gramaje et al. 2018; Moller and Kasimatis 1980).
Fungal entry points
Infection of grapevine by GTDs occurs after precipitation events, when spores of fungal pathogens (sexual and asexual) become airborne and colonize exposed pruning wounds resulting from winter pruning (Eskalen et al. 2007; Petzoldt et al. 1983; Rooney-Latham et al. 2005; Úrbez-Torres et al. 2008). Following pruning, grapevine wounds remain susceptible to infection by canker pathogens for several weeks (Eskalen et al. 2007; Munkvold and Marois 1995; Úrbez-Torres et al. 2008). During this time, the plants can become infected multiple times with one or more pathogens. Because several pathogens are frequently isolated from old cankers, GTDs are often referred to as a complex (Gramaje et al. 2018; Larignon and Dubos 1997; Rolshausen et al. 2004; Úrbez-Torres et al. 2006). Several years after infection occurs, grapevine yields can decline due to formation of wood cankers and/or plugging of the xylem and phloem vessels in the trunk or cordon, resulting in impaired translocation of water and nutrients. When the affected vineyards are no longer profitable, growers have no alternative but to remove the infected vines and replant.
Control of grapevine trunk diseases
Complete control of GTDs is virtually unattainable because of the high number of pruning wounds made on individual grapevines and the extended period of wound susceptibility, but there are options for minimizing the impact of these diseases. Previous studies evaluated a range of chemical fungicides for their ability to prevent infection of grapevine pruning wounds by E. lata. The most effective were carbendazim, tebuconazole, pyrimethanil, fluazinam and pyraclostrobin. However, these chemicals were applied to the wounds at rates greater than those currently registered for other diseases of grapevines (Sosnowski et al. 2008; Sosnowski et al. 2013), and some have been banned because of health and safety concerns (Bertsch et al. 2013).
Commercial chemical fungicide treatments available in the United States, such as a combination of Rally and Topsin M (active ingredients myclobutanil and thiophanate-methyl, respectively), have also been shown to be effective in controlling fungi associated with GTDs (Rolshausen and Gubler 2005; Sosnowski et al. 2008). In addition to chemical fungicides, biofungicides (for bio control without chemicals) have generated considerable interest as pruning wound protection strategies. Beneficial microorganisms are known to reside inside roots, stems and leaves of plants; these microorganisms help protect the plants from a range of infections (Lodewyckx et al. 2002). Biofungicides contain several of these beneficial microorganisms, including Trichoderma spp. and Bacillus spp., which have been shown to provide protection against GTDs (John et al. 2005; Kotze et al. 2011; Mutawila et al. 2016). In this study we sought to evaluate the efficacy of commercially available and experimental chemical and biological fungicides against E. lata and N. parvum infection on pruning wounds of grapevine under field conditions.
Field trial design
We conducted our field trial in an 8-year-old sauvignon blanc vineyard at the UC Davis Plant Pathology Field Station in Yolo County. Grapevines in the vineyard had been trained to bilateral cordons (the “arms” of the vine) on a horizontally divided trellis with typically 10 spurs per cordon. In early March 2019, we pruned the spurs so that each bore three, 1-foot-long buds. Within 24 hours of pruning, we sprayed liquid formulations of each treatment onto the exposed pruning wound until run off. The treatments were prepared according to commercial label recommendations (table 1) and applied with 1-liter handheld plastic spray bottles. Unless otherwise shown in table 1, the pruning wounds were artificially inoculated within 24 hours of spraying with a 20-μl aqueous spore suspension (approximately 1,000 spores per wound) of each fungal pathogen.
In this study, we inoculated a total of 10 grapevines per treatment. We applied a treatment combination (one pathogen + one fungicide) to two pruning wounds on each grapevine, organized in a random block design. We selected treatments based on varying groups or types of active ingredients and modes of action. In the control treatment, we applied sterile distilled water to wounds and then inoculated the wounds with the same spore concentration of each pathogen as we used in the fungicidal treatments.
Preparation of fungal inocula
We collected dead wood from grapevine trunks naturally infected with stromata of E. lata in a vineyard in Lodi, California. After releasing fruiting bodies (ascospores) from 5-centimeter-long wood segments (as described by Carter 1991), we adjusted the ascospore concentration to 2.5 × 104 ascospores/mL. We also collected dead grapevine wood with fruiting bodies (pycnidia) of N. parvum from naturally infected grapevine trunks in the same Lodi vineyard, and we adjusted the concentration to equal that of E. lata: 2.5 × 104 pycnidiospores/mL. We added a 0.05% solution of Tween 20 as a surfactant to both pathogen inocula. Prior to inoculation, we confirmed the viability of inoculum suspensions by plating on potato dextrose agar medium (PDA).
Fungal inocula were applied to pruning wounds following application of liquid treatments. Photo: Akif Eskalen.
Calculating efficacy of pathogen control
Treated spurs were allowed to stand for 6 months before collection and laboratory analysis. After we collected the spurs, we split each one with a knife longitudinally and then cultured six small tissue pieces (three from the pith and three from the margin of the dead wood or from any area exhibiting discoloration) on PDA amended with tetracycline at 100 mg/L (PDA-T). After incubating the tissues at room temperature for 7 to 14 days, we recorded the recovery of the fungal pathogens by means of their morphological characteristics. We confirmed the identity of isolates by sequencing the ITS region of the isolates' rDNA, and we calculated the efficacy of the protectants controlling the GTDs as the mean percent of infection (MPI). We used the following formula for the MPI calculation:
(Number of GTD-infected spur samples / number of total samples) × 100.
We calculated the mean percent disease control (MPDC) on the basis of the MPI of the inoculated control treatment, using the formula (100 × (1 − (MPI treatment / MPI inoculated control))). Means comparisons were made using Dunnett's test α = 0.05. We performed all data analysis using JMP software version 14 (SAS Institute, Cary, N.C.).
Differences in E. Lata and N. parvum responses
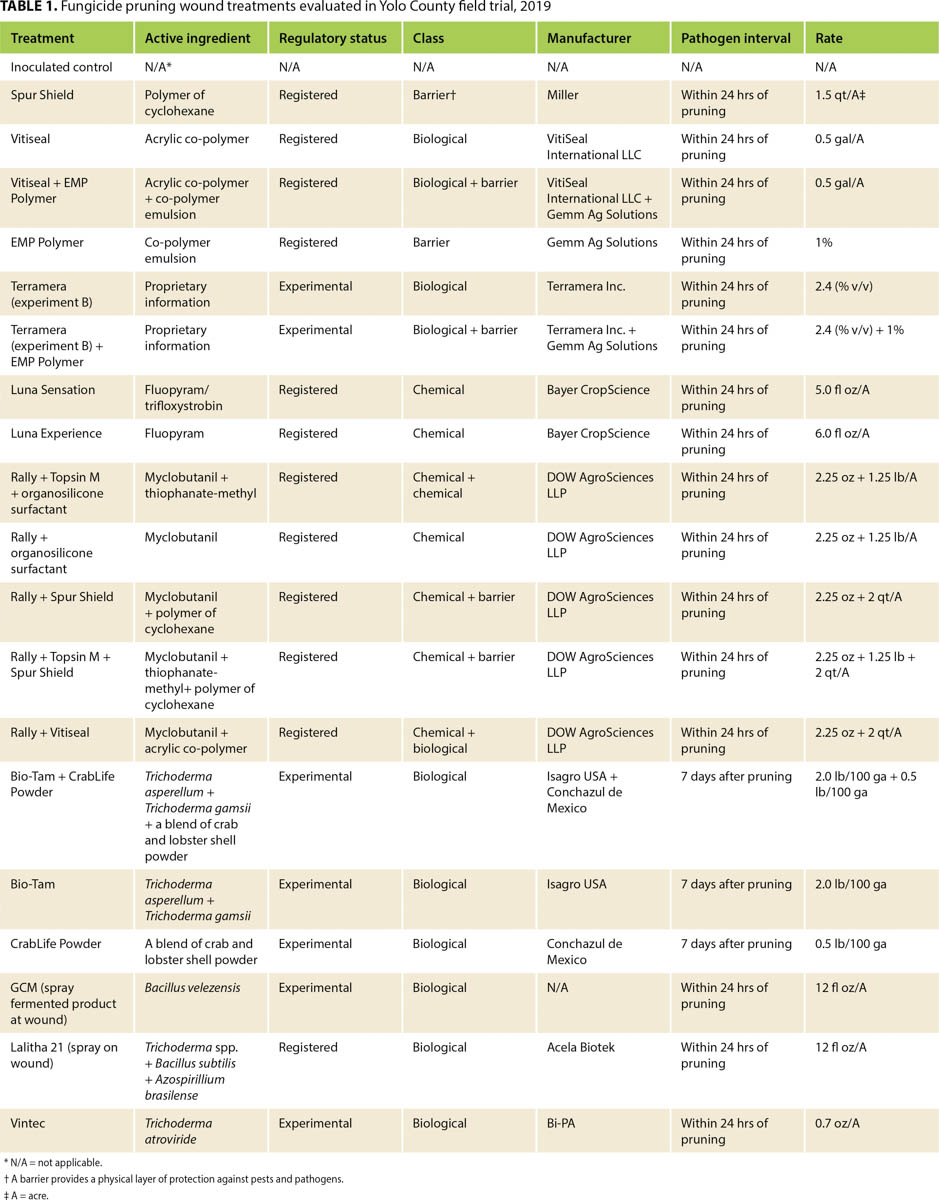
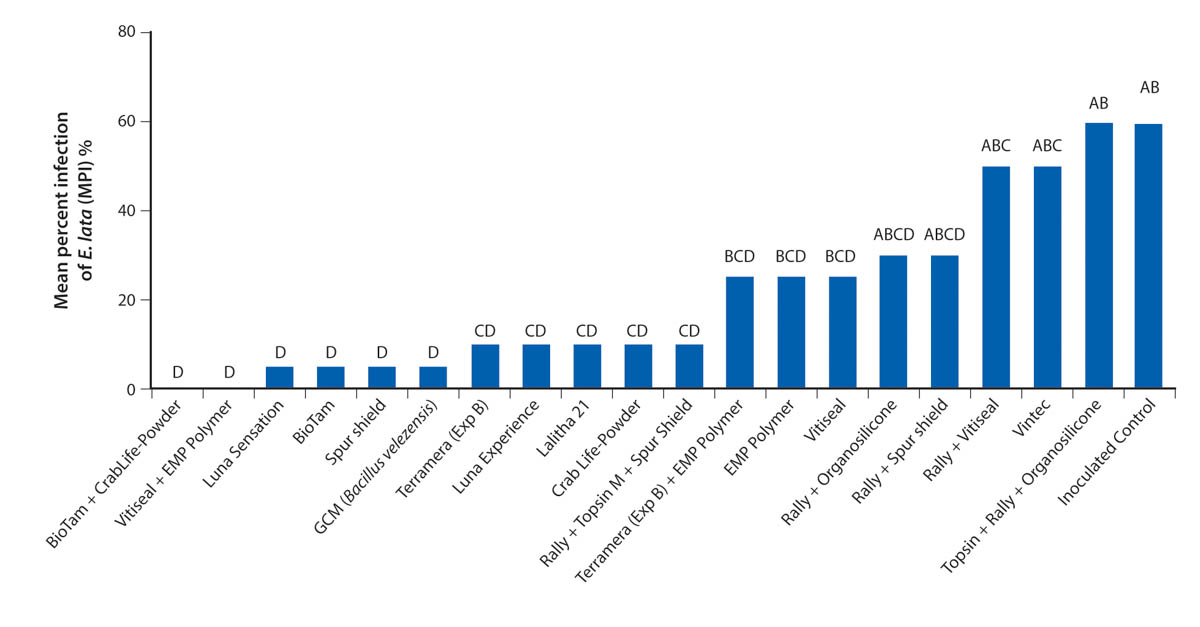
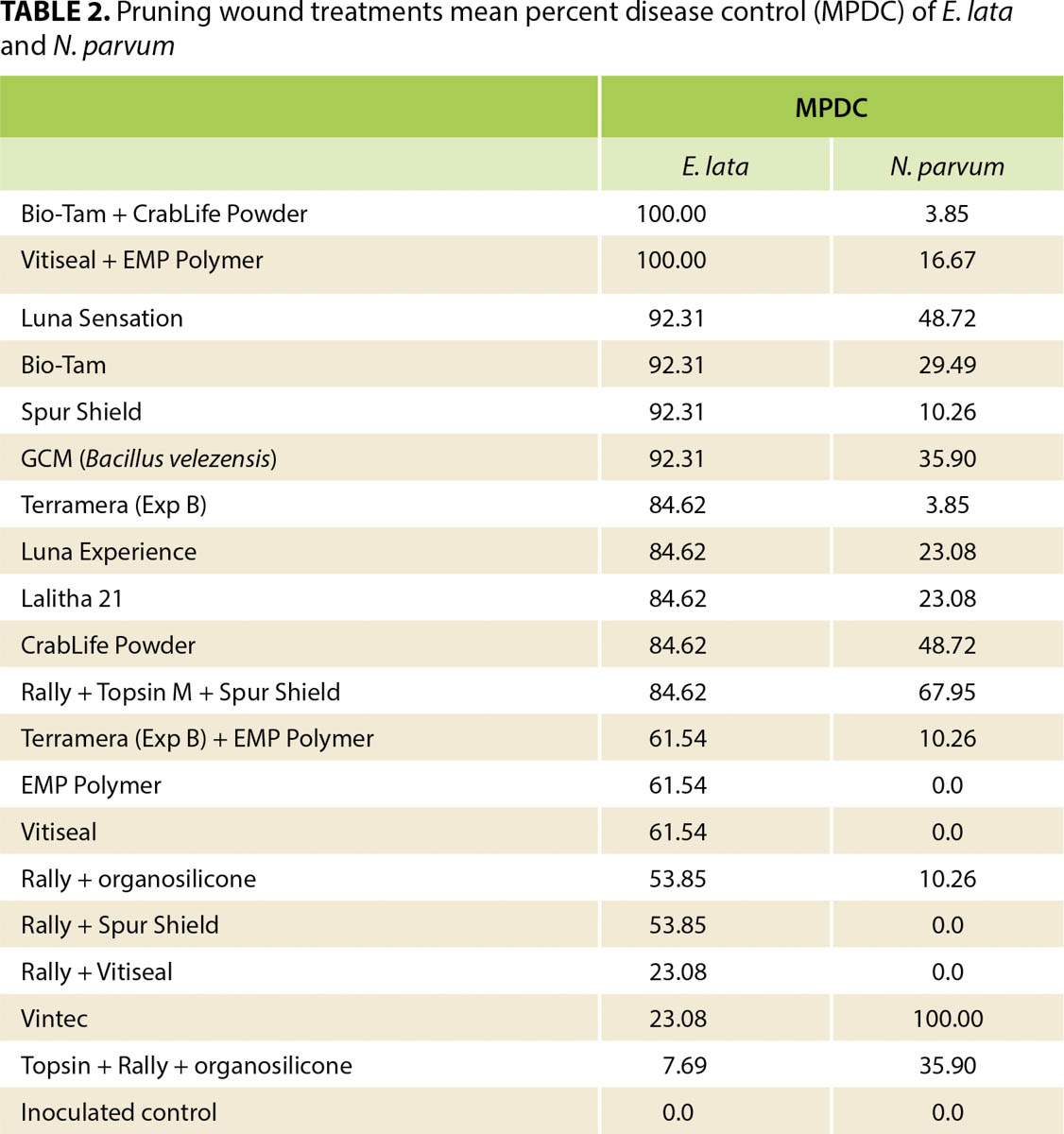
Both E. lata and N. parvum colonized the pruning wounds in our treated samples, but they had different infection rates (figs. 1 and 2). Treatments overall protected pruning wounds against E. lata infection more effectively than they protected against N. parvum. Of 20 treatments 16 controlled E. lata at least 50% more effectively than the inoculated controls did, and six of these treatments had an MPDC higher than 90% (table 2). For N. parvum, only Vintec and Rally + Topsin M + Spur Shield resulted in an MPDC of at least 50% (table 2). A total of eleven treatments limited the MPI of E. lata to under 20% and were significantly different (P < 0.05) from the MPI of the inoculated control treatment, but only Vintec and Rally + Topsin M + Spur Shield significantly reduced N. parvum infection compared to the inoculated control (figs. 1 and 2). The treatments EMP Polymer, Vitiseal, Rally + Spur Shield, and Rally + Vitiseal had an MPDC of 0% for N. parvum (table 2). These treatments in fact resulted in a higher MPI of N. parvum compared to that of the inoculated control, with the inoculated control yielding 78% MPI. The highest MPI for N. parvum was Rally + Vitiseal, yielding 90% (fig. 2). The low control rate of N. parvum versus E. lata can likely be attributed to N. parvum's more aggressive nature and faster colonization of woody tissue (Galarneau et al. 2015).
FIG. 1. Evaluation of pruning wound treatments mean percent infection (MPI) rates with E. lata located at UC Davis Plant Pathology Field Station, 2019. Bars represent the least square means of percent infection. Bars with a different letter are different according to Dunnett's test (P = 0.05).
FIG. 2. Evaluation of pruning wound treatments mean percent infection (MPI) rates with N. parvum located at UC Davis Plant Pathology Field Station, 2019. Bars represent the least square means of percent infection. Bars with a different letter are different according to Dunnett's test (P = 0.05).
Higher than natural pathogen pressure
To ensure significant infection of control spurs, we inoculated pruning wounds with a significantly higher number of spores than that estimated to infect a pruning wound naturally (Carter and Moller 1971). Sosnowski and Mundy (2019) showed that MPI from noninoculated controls was 3% to 6% for E. lata, and 12% to 17% for N. luteum. In contrast, the MPI for our inoculated controls was 65% for E. lata and 78% for N. parvum (fig. 2). Similar results were reported by other researchers using high spore-count treatments. Rolshausen et al. (2010) reported that the inoculated control treatment for a Botryosphaeriaceae pathogen had an MPI of 90%, and Sosnowski and Mundy (2019) reported an MPI of 59% for E. lata averaged over multiple years. The high MPI rates in our inoculated controls are therefore likely the result of the large spore concentrations we applied to the pruning wounds. In future studies, it may be prudent to compare the effectiveness of treatments under both high and moderate-to-low disease pressure. For example, Sosnowski and Mundy (2019) in New Zealand showed that when disease pressure was moderate (i.e., 40% to 60% recovery of pathogens from inoculated controls), fungicide efficacy was far greater than when the disease pressure was high (greater than 60%). Under the high disease pressure in our study (greater than 60%), the majority of treatments performed well as pruning wound protectants against E. lata, but this might not be the case under natural conditions.
Trichoderma-based biological fungicides
In this study, Trichoderma-based biological fungicides performed the best against both of the fungal pathogens we investigated, with Vintec performing the best against N. parvum and Bio-Tam + CrabLife Powder performing the best against E. lata (figs. 1 and 2). It may be prudent in future studies to combine Vintec and Bio-Tam + CrabLife Powder in a single application to see if they can provide effective pruning-wound protection against multiple GTD pathogens. Interestingly, Bio-Tam was more effective than Bio-Tam + CrabLife Powder against N. parvum, which suggests that CrabLife Powder might have an inhibitory effect on the Trichoderma spp. that are the active ingredients in Bio-Tam. The commercial success of biocides containing Trichoderma spp., representing more than 60% of registered biocides, is based on the benefits they confer. Depending on the strain, Trichoderma species can stimulate plant growth, suppress pathogens by direct competition for nutrients and space, exhibit antibiosis and induce systemic resistance (Harman 2006; Mukherjee et al. 2013). Other pruning-wound studies have also shown that Trichoderma spp. can provide effective control against fungal pathogens associated with GTDs (Berbegal et al. 2020; John et al. 2005).
Simultaneous control of pathogens is necessary
Grapevine spur positions were pruned to three 1-foot-long buds in early March 2019. Photo: Akif Eskalen.
Identifying pruning wound treatments that can control multiple GTD pathogens will be essential for mitigating GTD infections. In our study, some treatments provided effective control of both E. lata and N. parvum, including Luna Sensation, Rally + Topsin M + Spur Shield, CrabLife Powder, Bio-Tam and GCM (Bacillus velezensis). Rally + Topsin M + Spur Shield had a high efficacy against both E. lata (MPDC of 84.62%), and N. parvum (MPDC of 67.95%). While we found that Trichoderma-based biological fungicides are capable of protecting wounds with high efficacy, it should be highlighted that some biofungicides in this trial were not effective in controlling both pathogens. For example, Bio-Tam + CrabLife Powder had 100% disease control of E. lata, yet this combination only had 3.85% MPDC of N. parvum (table 2). Inversely, Vintec had 100% disease control of N. parvum but only 23% MPDC of E. lata. Similar results were reported by Rolshausen et al. (2010) where biopaste was very efficient in controlling E. lata but did not control species in the Botryosphaeriaceae family.
Testing of various cultivars and pathogens needed
While our results are encouraging, the climatic conditions at our experimentation site and the cultivar we used for this study likely differ from those of other grapevine-growing regions. Further research should evaluate these promising fungicides against GTDs pathogens in diverse conditions, including various geographical locations and cultivars. The susceptibility of pruning wounds to fungal pathogens may also vary depending on the fungal isolate used in a specific study (Sosnowski et al. 2007). Future studies on fungicide efficacy should include multiple isolates of a specific pathogen species.
In conclusion, our study has shown that several chemical and biological fungicides can provide efficient protection of pruning wounds against at least one canker pathogen, and some are effective at controlling both. The combination of Luna Sensation with Rally + Topsin M + Spur Shield, for example, offered simultaneous control of both E. lata and N. parvum. Remaining challenges include improving accurate diagnoses of GTDs and identifying additional treatment products efficacious against a broad diversity of fungal canker pathogens.