All Issues
Growth stage influences level of resistance in glyphosate-resistant horseweed
Publication Information
California Agriculture 61(2):67-70. https://doi.org/10.3733/ca.v061n02p67
Published April 01, 2007
PDF | Citation | Permissions
NALT Keywords
Abstract
While glyphosate-resistant horseweed has not previously been reported in California, we suspected that it might exist, especially in noncrop areas. We collected horseweed seeds from two locations in the San Joaquin Valley and treated greenhouse-grown plants at different stages with different amounts of glyphosate. This study showed that a glyphosate-resistant biotype of horseweed exists in the noncrop areas of Dinuba, in Tulare County, and that the level of resistance may be influenced by the plant's growth stage at the time of glyphosate application.
Full text
Horseweed, or marestail (Conyza canadensis L. Cronq.; Asteraceae family), is an annual, native, North American plant, which often colonizes roadsides, fallow fields, fencerows and the nontilled rows of perennial crops in the San Joaquin Valley. Although horseweed has been in California for a long time, increased invasions of this weed have been observed in orchards and vineyards in the southern San Joaquin Valley over the past 5 years.
In the past, horseweed was adequately controlled by pre- and post-emergence herbicide treatments, fall and/or spring cultivation, or hand-pulling in perennial tree and vine crops. However, in recent years this weed has become more difficult to control. This may be because of emerging air-quality regulations that have led to restrictions on agricultural cultivation near urban areas, and water-quality concerns that have led to restrictions on the use of certain preemergence herbicides in the San Joaquin Valley. Furthermore, in years when commodity prices for crops such as raisins and stone fruits were low, weed control efforts were reduced in vineyards and orchards, and horseweed densities increased.
A glyphosate-resistant horseweed plant shows the regrowth of new tissue 3 to 4 weeks after glyphosate was applied.
Growers who choose chemical weed control face challenges in the San Joaquin Valley. Many vineyards, orchards and noncrop areas (such as canal banks) in this region are located in Ground Water Protection Areas, state-designated areas that are vulnerable to pesticide leaching or runoff into groundwater (DPR 2004). Several preemergence herbicides, including simazine, that are effective on horseweed are now severely restricted in Ground Water Protection Areas. Instead, relatively inexpensive, broad-spectrum, postemergence herbicides — primarily glyphosate (Roundup) — are often used in these areas. (Postemergence herbicides are applied on the weeds after they emerge and thus they do not leach into the groundwater, as opposed to preemergence herbicides that are applied to the soil and incorporated.)
However, in various parts of the world horseweed has developed resistance to broad-spectrum herbicides such as the triazines (Gressel et al. 1982; Heap 2007), paraquat (Smisek et al. 1998), the acetolactate synthase inhibitors (Heap 2007) and glyphosate (VanGessel 2001). The first case of a glyphosate-resistant horseweed in North America was reported in Delaware in 2000 (VanGessel 2001). Since then, 13 other states have reported glyphosate-resistant horseweed (Heap 2007). All of these reports were from annual row-crop systems such as cotton (Gossypium sp.) and soybean (Glycine max). Repeated use of the same herbicide is the main reason that weeds develop herbicide resistance worldwide (Holt 1992). Researchers believe that the intensive use of glyphosate in crops that are genetically engineered to be resistant to this herbicide has resulted in the selection of weed populations that are also naturally resistant (Nandula et al. 2005). Although there are reports of glyphosate-resistant horseweed in perennial cropping systems in Brazil and China (Heap 2007), there have been no reports of glyphosate-resistant horseweed in perennial cropping systems or noncrop areas in North America.
Glyphosate was applied at increasing rates (0, 1, 2 and 4 pounds active ingredient per acre) to resistant horseweed plants from Dinuba, Tulare County (left) and susceptible plants from Fresno County (right).
A field manager in Dinuba, Tulare County, recently reported poor control of horseweed with glyphosate on an irrigation canal bank. Glyphosate had been used repeatedly at this site during the previous several years, and we suspected glyphosate resistance after observing plants that escaped treatment and those that were controlled by glyphosate at this site. Preliminary studies at this site and in the greenhouse showed that while horseweed seedlings were effectively controlled by glyphosate at a rate of 4 pounds active ingredient per acre (ai/ac), about 40% of the horseweed at the 18- to 21-leaf rosette stage did survive. We decided to test the Dinuba horseweed for glyphosate resistance and evaluate how glyphosate rate and plant growth stage affect resistance.
Testing for glyphosate resistance
We collected horseweed seeds from the suspected glyphosate-resistant (GR) population in Dinuba (36°29'15” N; 119°24'10” W) and from a population in western Fresno County believed to be glyphosate-susceptible (GS), where effective control had been obtained with a labeled rate of glyphosate (control). Seeds were collected in fall 2004 and stored at room temperature (70°F). The experiment was done twice, first with seeds planted in early spring and second with seeds planted in late summer. The first experiment was conducted from April 6 to Sept. 4, 2005, and the second from Aug. 10, 2005, to Jan. 3, 2006.
Seeds for this study were collected from horseweed plants that had survived glyphosate applications along a canal bank in Dinuba, Tulare County.
Horseweed seeds were planted in plastic germination trays in the lab and moved to a greenhouse following emergence. The greenhouse temperatures were set at 75°F to 80°F during the day and 60°F to 65°F at night. No supplemental lighting was used. When the seedlings developed two to three leaves, they were transplanted into plastic pots (6 inches deep and 4 inches wide) containing a commercial potting mix. For the purpose of this study, seeds from the Dinuba and west Fresno County sites were designated as GR and GS horseweed biotypes, respectively.
The experimental design was a two-factor, completely randomized block with five replications. The two factors included five glyphosate application timings based on horseweed growth stage (5 to 8 true leaves, 11 to 15 true leaves, 18 to 21 true leaves, bolting to 6 inches tall, and 6.1 to 12 inches tall) and four rates of glyphosate (0, 1, 2 and 4 pounds ai/ac). A fully expanded leaf was considered a true leaf. Four extra plants (two GR and two GS) were included in each replication at 18 to 21 true leaves (rosette stage) for glyphosate treatments of 8 and 16 pounds ai/ac. These extra plants were included because growers and land managers generally treat horseweed at the rosette stage in early spring in the San Joaquin Valley. There were 44 pots containing either the GR or GS biotype for each of the five growth stages, for a total of 220 pots. The plants were watered regularly and fertilized twice during the growing season with a commercial fertilizer (MiracleGro).
Glyphosate, formulated as Roundup Weathermax (5.5 pounds ai/gallon), was used in the study. No additional surfactants were added to the spray solution. Treatments were applied at the designated growth stage with a carbon dioxide backpack-sprayer. The spray was discharged 18 inches above the target plants through a 40-inch boom with a single flat-fan nozzle (TeeJet XR8002EVS) in the center and a blank at each end. The system was pressurized to 30 pounds per square inch (psi) to deliver the herbicide solution at 35 gallons per acre (gpa; broadcast acre basis) in a 20-inch band. The plants were moved outside the greenhouse, sprayed and moved back into the greenhouse. The mortality of each plant was evaluated weekly, and classified as “alive” or “dead.” Plants were designated as dead when the above-ground plant parts disintegrated and showed no traces of green tissue.
In the first experiment, survivors were allowed to grow until anthesis (the period during which a flower is fully open) of the first flower, and then the aboveground biomass was collected. In the second experiment, biomass was collected prior to flowering due to a severe aphid infestation. In both experiments, plants were clipped at the surface of the soil, placed in separate paper bags, dried to constant weight in a forced-air oven at 140°F, and their dry weights recorded.
Problematic horseweed infestations have become more common among perennial crops in the San Joaquin Valley, such as near this Fresno County raisin vineyard. Treatments should be applied soon after horseweed emerges, before plants develop more than eight true leaves.
Mortality and shoot biomass data were subjected to analysis of variance using GLM procedures in SAS with an alpha level of 0.05. Mortality data for both experiments were combined because there were no interactions (P > 0.05) between experiment and biotype or between experiment and glyphosate rate for plant mortality. Shoot biomass data was analyzed separately for the two experiments because of differences in the development stage of the plants at the time of harvest.
Mortality of the GS and GR horseweed plants at the rosette stage was regressed against glyphosate rate using a nonlinear sigmoidal dose-response model in SigmaPlot: γ = min. + max. - min./1 + 10(LD50 - x) [1], where γ is plant mortality, min. is the minimum response limit (the minimum dose required for plant mortality among a group of plants), max. is the maximum response limit, LD50 is the herbicide rate to achieve 50% mortality, and x is the concentration of glyphosate rate.
Growth stage and mortality
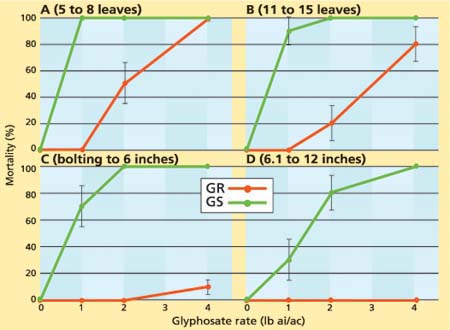
While Koger et al. (2004) found that the growth stage of horseweed had little effect on the level of glyphosate resistance, our data indicated that the level of resistance is influenced by the growth stage at the time of glyphosate application. At the 5- to 8-leaf stage, all of the GR horseweed plants survived the 1-pound glyphosate treatment (fig. 1A). However, only half survived the 2-pound treatment, while none survived the 4-pound treatment. At this stage, none of the GS plants survived any of the glyphosate treatments (fig. 1A).
At the 11- to 15-leaf stage, all of the GR horseweed plants survived the 1-pound glyphosate treatment while only 20% survived the 4-pound treatment (fig. 1B). In contrast to the 5- to 8-leaf stage, 10% of the GS plants at the 11- to 15-leaf stage survived the 1-pound glyphosate treatment (fig. 1B). After the plants bolted, most of the GR plants survived the 4-pound glyphosate treatment (figs. 1C, 1D). Similarly, delaying glyphosate application until bolting increased the chances of survival for GS plants, as 30% of them survived the 1-pound treatment (fig. 1C). Tolerance of GS plants to glyphosate further increased when the herbicide was applied at the 6.1- to 12-inch stage, as 70% and 20% of the plants survived the 1- and 2-pound treatments, respectively (fig. 1D).
Fig. 1. Percentage mortality of glyphosate-resistant (GR) and glyphosate-susceptible (GS) horseweed plants under different glyphosate rates sprayed at the (A) 5- to 8-leaf, (B) 11- to 15-leaf, (C) bolting to 6-inch and (D) 6.1- to 12-inch stages.
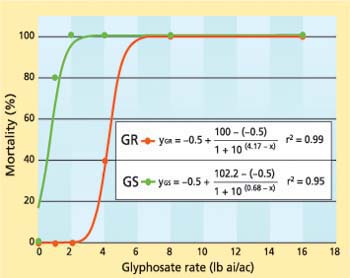
Fig. 2. Nonlinear regression of percentage mortality of glyphosate-resistant (GR) and glyphosate-susceptible (GS) horseweed plants as a function of different glyphosate rates sprayed at the 18- to 21-leaf (rosette) stage.
Glyphosate resistance in horseweed is believed to be due to the limited translocation of glyphosate to the roots and growing points of the plant (Feng et al. 2004). This could also be the reason for the increased glyphosate resistance that we found at later growth stages in the GR plants.
Based on a nonlinear regression model predicting percentage mortality as a function of herbicide rate, the glyphosate treatments required to kill 50% of the GR and GS plants at the rosette stage were 4.17 and 0.68 pounds, respectively (fig. 2). We found that at the rosette stage, 20% of the GS plants survived the 1-pound treatment and none survived the higher treatments, while none of the GR plants survived the 8- or 16-pound treatments. Therefore, based on the model, the GR plants were approximately six times more resistant to glyphosate than the GS plants at the rosette stage. Similarly, VanGessel (2001) reported an 8- to 13-fold increase in glyphosate resistance in GR horseweed biotypes from Delaware, and Koger et al. (2004) reported an 8- to 12-fold increase in glyphosate resistance in GR horseweed biotypes from Mississippi.
Plant biomass effects
The GR plants from Dinuba generally grew bigger than the GS plants from Fresno County. A t-test for the control treatment (no glyphosate) showed that at the time of flowering, the GR plants had 31% more above-ground biomass than the GS plants in the first experiment, and 27% more in the second experiment.
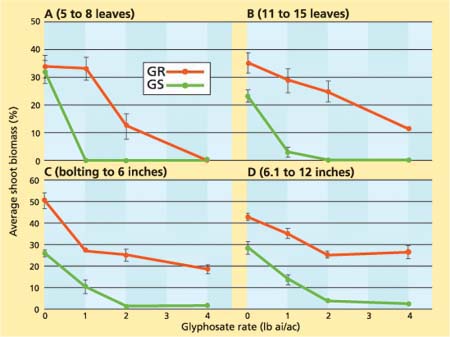
Fig. 3. Average shoot biomass (± SE) of glyphosate-resistant (GR) and glyphosate-susceptible (GS) horseweed plants in experiment 1 as a function of different glyphosate rates sprayed at the (A) 5- to 8-leaf, (B) 11- to 15-leaf, (C) bolting to 6-inch and (D) 6.1- to 12-inch stages.
Higher glyphosate levels generally reduced the shoot biomass of the GR plants that survived the herbicide application (figs. 3A-D). For example, shoot biomass was reduced 18%, 30% and 67% by the 1-, 2- and 4-pound glyphosate treatments, respectively, when the herbicide was applied at the 11- to 15-leaf stage (fig. 3B). When glyphosate was applied at the 5- to 8-leaf stage, some GR plants survived the 2-pound treatment, but they were stunted and accumulated very little biomass compared to the untreated control plants (fig. 3A). In addition, the shoot biomass of the GR horseweed was greater than that of the GS horseweed even under nonsprayed conditions (figs. 3B-D). Although a few GS plants escaped the 1- and 2-pound treatments when glyphosate was applied postbolting, their shoot biomass was reduced up to 92% (fig. 3D). Similar reductions in shoot biomass of GR plants were also observed in the second experiment as glyphosate rates increased (data not shown).
Resistance and horseweed control
In these experiments, most of the GR horseweed plants that initially appeared to be dead began growing again approximately 3 to 4 weeks after glyphosate application, producing green leaves from the center of the rosette and starting to reaccumulate shoot biomass. Although the biomass of the horseweed was reduced in our study, all the surviving plants still produced flowers in the first experiment. We found that the GR plants from Dinuba amassed more aboveground biomass than those from Fresno County and appeared more vigorous. However, it is difficult to associate glyphosate resistance with plant vigor. A comparative study of progeny of GR and GS plants derived from the same parents may be needed to verify if there are any fitness or plant vigor costs associated with glyphosate resistance in horseweed.
Our results showed that the horseweed from Dinuba was resistant to glyphosate, but that the level of resistance varied with growth stage. GR plants from Dinuba could likely be controlled at the 5- to 8-leaf stage with 2-and 4-pound glyphosate treatments. At later stages, even some of the GS horseweed from Fresno County escaped the lower rates of glyphosate. These results highlight the importance of controlling weeds at an early growth stage.
When a postemergence herbicide such as glyphosate is used for horseweed control, it is important to apply the treatment soon after the horseweed emerges, preferably before plants develop more than eight true leaves. This may result in complete control of GS and partial control of GR plants. The application of glyphosate to horseweed during or after the bolting stage can result in some escapes of GS plants and no control of GR plants. If the horseweed population is to be reduced, several successive postemergence herbicide applications are needed to control plants that may emerge in multiple flushes over the growing season. This could result in further increase in herbicide use in Ground Water Protection Areas and perennial cropping systems that rely solely on postemergence weed control.
In order to prevent or delay the onset of herbicide resistance in horseweed, an integrated program should be developed to manage this plant, particularly in noncrop areas, orchards and vineyards located in Ground Water Protection Areas of the San Joaquin Valley. Similarly, a resistance management strategy must also be adopted for this weed in glyphosate-resistant crops. Although the difference in the intra-and interspecific competitive ability of the two biotypes is unknown, it may be beneficial to prevent the spread of glyphosate-resistant Dinuba-type horseweed because this biotype appears to be more vigorous than the glyphosate-susceptible Fresno County biotype.