All Issues
Various novel insecticides are less toxic to humans, more specific to key pests
Publication Information
California Agriculture 59(1):29-34. https://doi.org/10.3733/ca.v059n01p29
Published January 01, 2005
PDF | Citation | Permissions
Abstract
A number of novel insecticides have recently been registered for insect control in agriculture. A major advantage of these new products is that they act on insect biological processes that humans do not experience, such as molting. Many also have greater selectivity to target specific species, so they are less likely to harm natural enemies when compared with the broader spectrum organophosphate, carbamate, neonicotinoid and pyrethroid insecticides. Such novel insecticides currently in use include four targeting lepidopteran pests, three targeting sucking insects, one specific to dipteran leafminers and one insect growth regulator that controls a wide range of insects. One negative aspect of these insecticides is that because of their narrower range of activity — controlling only a limited number of pests — growers may need to apply additional pesticides for secondary pest groups that have poor biological control, increasing the total number of treatments per acre and total pest-control costs.
Full text
A number of novel insecticides with unique modes of action were registered during the late 1990s and early 2000s for insect control in agriculture. These new insecticides have several advantages over older classes of insecticides. First, most of the products in this group act on insect processes that humans do not experience, such as molting. Low mammalian toxicity allows for short re-entry and preharvest intervals, allowing the insecticides to be easily incorporated into pest control programs. Many also have greater selectivity and so are less likely to harm natural enemies than the broad-spectrum organophosphate (OP), carbamate, neonicotinoid and pyrethroid insecticides. As such, they are less likely to cause outbreaks of secondary pests that are well controlled by natural enemies, and may be used as “clean-up” sprays to manage outbreaks of pests caused by broad-spectrum insecticides. The registration of these insecticides has helped to greatly reduce OP and carbamate insecticide use in California. This has had an especially significant impact in cotton, citrus and stone fruits, where OP and carbamate use has been reduced by as much as 70% since the late 1990s.
While often effective at controlling specific pests, less-toxic new insecticides cab also have unintended impacts. When pyrlproxyfen was sprayed to control red scale in citrus, it also caused gross abnormalities in vidalia beetle pupae, left (normal) and right (abnormal) . Vidalia beetles, inset (adult stage), are predators needed to control cottony cushion scale; as a result, secondary outbreaks of scale occurred.
The new insecticides also have some disadvantages. Because of their narrower range of activity, each insecticide generally controls only one pest group within a crop. The grower may need to apply additional insecticides for secondary pests that have inadequate natural control, increasing the total number of treatments per acre and total pest-control costs. In addition, many of the novel insecticides have fairly short residual activity or affect only immature stages of insects, so the treatment timing is less flexible compared with broad-spectrum insecticides. Finally, the cost of the new insecticides is usually significantly higher than the older products.
It is fortuitous that in recent years insecticides from different chemical classes have been registered to control lepidopteran (primarily moths) and homopteran (primarily scales and white-flies) pests, because many insects in these groups have developed resistance to the older pesticides. The simultaneous registration of insecticides with unique modes of action allows growers to alternate the insecticides used, reducing the rate at which resistance develops. Insecticide resistance in key pests will continue to be a major impetus for adopting novel insecticides.
Insecticides for Lepidoptera
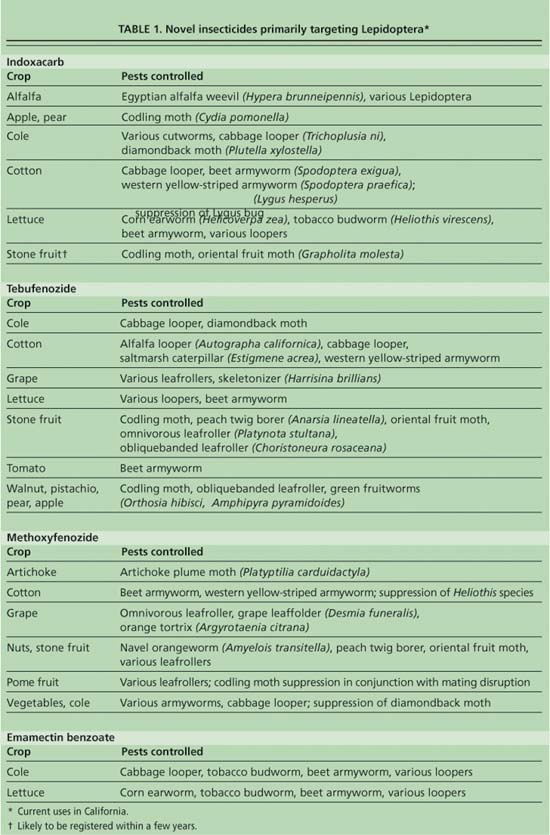
Four insecticides that have activity primarily affecting lepidopteran pests — indoxacarb (Avaunt, Steward), tebufenozide (Confirm), methoxyfenozide (Intrepid) and emamectin benzoate (Denim, Proclaim) — are registered for a number of crops in California. The greatest uses of these insecticides are in cotton, cole crops, lettuce, nuts, and stone and pome fruits (table 1).
In stone fruit, the use of these insecticides — in combination with Bacillus thuringiensis (Bt) products, spinosad and mating disruption during the growing season — has greatly reduced the need for dormant sprays of OP, carbamate and pyrethroid insecticides for peach twig borer (Anarsia lineatella). This has benefited the stone fruit industry by reducing pesticide residues in surface water, by preserving natural enemies needed for other pests such as San Jose scale (Diaspidiotus perniciosus), and by reducing secondary outbreaks of spider mite pests caused by broad-spectrum-insecticide disruption of their natural enemies.
The Central Coast Vegetable Integrated Pest Management Program for pest management in lettuce provides another example of the significant role that these narrow-spectrum insecticides play in Lepidoptera control. Lettuce is highly susceptible to pest damage at the seedling stage and during head formation. Many of the seedling pests — such as crickets, flea beetles, aphids and whiteflies — are controlled with broad-spectrum OP, carbamate, pyrethroid or neonicotinoid insecticides. These insecticides reduce or eliminate the natural enemies that attack the lepidopteran pests, sometimes causing outbreaks; selective insecticides help to bring the Lepidoptera back under control without creating additional problems.
In addition, there are a number of lepidopteran pests that attack both head and leaf lettuce, including cabbage looper (Trichoplusia ni), beet armyworm (Spodoptera exigua), corn earworm (Helicoverpa zea) and tobacco budworm (Heliothis virescens). Lepidopteran pests can destroy seedlings, bore holes and leave frass or insect body contaminants throughout the growth cycle of the lettuce, necessitating multiple treatments. Indoxacarb and tebufenozide provide unique, selective chemistries for these pests and act as rotational alternatives to each other, as well as spinosad and Bt, helping to reduce the rate that insecticide resistance develops.
Indoxacarb
Indoxacarb is an oxadiazine insecticide that blocks the sodium channels in insect nerve cells, causing lepidopteran larvae to stop feeding within 4 hours, become paralyzed and die within 2 to 5 days (McCann et al. 2001). It is more effective as a stomach poison than as a contact poison. Indoxacarb is fairly selective, having activity primarily against lepidopteran larvae and certain species of sucking insects such as Lygus bugs. However, the activity of indoxacarb against the sucking insects is weaker than for Lepidoptera because of its slower bioactivation, lower sensitivity and a less favorable method of oral uptake in the sucking insects. Indoxacarb allows most predators and immature wasp parasites to survive (Hewa-Kapuge et al. 2003; Studebaker and Kring 2003). However, the wet residues of indoxacarb are toxic to bees and adult wasp parasites.
Indoxacarb controls important pests in alfalfa, apples, cole crops, cotton, lettuce and pears. Populations of obliquebanded leafroller (Choristoneura rosaceana) in Michigan (Ahmad et al. 2002) have exhibited resistance to indoxacarb in regions where it has not been used, suggesting cross-resistance to older groups of insecticides. This emphasizes the need for the rotation of indoxacarb with emamectin benzoate, the dibenzoylhydrazine insect growth regulators (IGRs) and other insecticides to maintain the efficacy of all groups of insecticides.
Tebufenozide
Tebufenozide is a dibenzoylhydrazine stomach poison that acts as an IGR specifically for Lepidoptera. It mimics a molting hormone and blocks the completion of the normal molting process (Retnakaran et al. 2001). The insect stops feeding within a few hours and undergoes a premature lethal molt within 3 to 7 days, becoming trapped within the shedding head capsule. Tebufenozide must be ingested to take effect and is thus slow-acting, with a residual activity of 14 to 21 days. Application timing is critical, because it is more active on early larval stages (Waldstein and Reissig 2001). It is non-toxic to honeybees and is selective, not affecting most natural enemies (Dhadialla et al. 1998).
The crops for which tebufenozide is currently registered include cole crops, cotton, grapes, lettuce, tomatoes and some nuts, pome and stone fruits. Low levels of resistance to tebufenozide have been found in codling moth (Cydia pomonella), beet armyworm and obliquebanded leafroller populations that were not exposed to this insecticide (Moulton et al. 2002; Ahmad et al. 2002), suggesting that there may be cross-resistance to older classes of compounds, including OPs. There is also likely to be crossresistance between tebufenozide and methoxyfenozide, because they have the same mode of action. These insecticides will need to be used infrequently, alternated with other insecticide chemistries, and coupled with alternative methods of control, such as mating disruption, to delay resistance in key pests.
Because lettuce is highly susceptible to insect damage at the seedling stage, many growers spray broad-spectrum insecticides. Some newer, more selective insecticides can bring lepidopteran pests under control without hurting their natural enemies and causing secondary pest outbreaks.
Methoxyfenozide
Methoxyfenozide is a dibenzoylhydrazine IGR, similar to tebufenozide in its mode of action, its ability to induce a lethal molt and its specificity for Lepidoptera (Carlson et al. 2001). Methoxyfenozide was only recently (2003) registered in California and its use is likely to increase due to its better binding with lepidopteran receptors and longer residuality compared with tebufenozide. Methoxyfenozide has a much lower ability to bind with receptors in nonlepidopteran species, making it a highly selective insecticide and useful in a number of crops. Low levels of resistance to methoxyfenozide in codling moth, beet armyworm and obliquebanded leafroller have been found, necessitating prevention precautions similar to those for tebufenozide.
Emamectin benzoate
Emamectin benzoate is a second-generation avermectin analog with exceptional activity against lepidopterans, acting by decreasing the excitability of neurons. Shortly after contact or feeding exposure, the insect larvae stop feeding, become irreversibly paralyzed and die in 3 to 4 days. Emamectin benzoate toxicity is broader spectrum than methoxyfenozide, tebufenozide or indoxacarb, which is a benefit in that it kills a wide variety of lepidopterans (Argentine et al. 2002). However, its broad-spectrum activity also makes fresh residues toxic to natural enemies (Studebaker and Kring 2003). Natural enemy survival improves after about 5 days due to rapid photodegradation. The toxic activity lasts longer for the pest because the photodegradate moves through plant tissue (translaminar activity) and is toxic to the plant-feeding pest.
Emamectin benzoate is used primarily against pests in cole crops and lettuce. (It is registered for cotton in other states, but not California.) Emamectin benzoate provides a rotational insecticide for the control of caterpillars and so helps to reduce the development of resistance. Populations of obliquebanded leafroller tested with emamectin benzoate have shown only slight levels of resistance (Waldstein and Reissig 2001).
Treatments for sucking insects
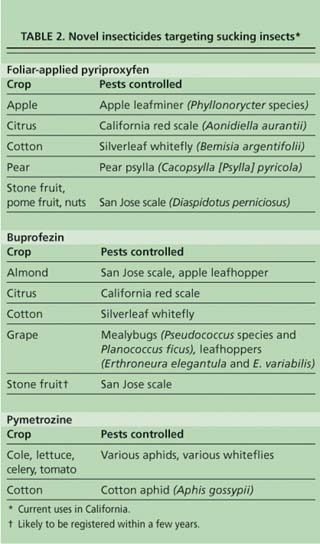
Three insecticides — pyriproxyfen (Esteem, Knack, Seize), buprofezin (Applaud, Courier) and pymetrozine (Fulfill) — have activity primarily affecting sucking insect pests such as whiteflies and armored scales, many of which have developed resistance to OP, carbamate or pyrethroid insecticides. The greatest uses of these insecticides for whitefly control are in cotton and tomatoes, for California red scale (Aonidiella aurantii) in citrus, and for San Jose scale in nuts, and stone and pome fruits (table 2). Their cost is often significantly higher than OPs, carbamates and pyrethroids.
Pyriproxyfen
Pyriproxyfen is a pyridine compound that acts as a juvenile hormone mimic IGR, inhibiting egg production and the metamorphosis of immature stages into adults (Ishaaya et al. 1994). It is most effective in late-stage larvae or nymphs and early pupal stages when juvenile hormone is normally low. It is active primarily against sucking insects such as scales, pear psylla (Cacopsylla [Psylla] pyricola) and whiteflies. It is also effective against fire ants and apple leafminers (Phyllonorycter species). Because of its persistence and efficacy, pyriproxyfen has been extremely effective in reducing California red scale and San Jose scale populations that developed resistance to OP insecticides. It is safer for hymenopterous parasites than OP insecticides, greatly increasing parasite numbers and so improving the control of both target and nontarget pests within crop systems. Pyriproxyfen is toxic to crustaceans, limiting its use around bodies of water.
Citrus provides an example of two potential problems associated with the use of IGR insecticides in agriculture. First, pyriproxyfen is not fully compatible with natural enemies because it is highly toxic to predatory coccinellid beetles, halting both egg hatch and pupation (Grafton-Cardwell and Gu 2003). The heavy use of pyriproxyfen in citrus for California red scale control created a cottony cushion scale (Icerya purchasi) problem due to the loss of vedalia beetle (Rodolia cardinalis). It may also be responsible for pest resurgences in other crops due to its effect on coccinellid predators of armored scales, mealybugs and mites.
Second, pyriproxyfen has a narrower range of activity compared with the carbamate and OP insecticides. Pyriproxyfen has no effect on the secondary pests forktailed bush katydid (Scudderia furcata) or citricola scale (Coccus pseudom-agnoliarum), which lack natural enemies, and so these insects have become primary pests that must be controlled with additional insecticide treatments. In past years, katydids and citricola scale were easily controlled by the OP treatments applied for California red scale.
Currently, pyriproxyfen is used in California to control pests in apples, citrus, cotton, nuts, pears, and stone and pome fruits. In bait form, it is effective against the protein-feeding native southern fire ant (Solenopsis xyloni). An extensive resistance-management program has been developed for cotton that limits the number of applications of pyriproxyfen to one per growing season to reduce the rate of resistance development. Caution should be exercised by growers as to the frequency of application, because resistance has begun to develop in whitefly populations in other areas of the world, even when the number of applications per season was limited to one (Horowitz et al. 2002).
Buprofezin
Buprofezin is a thiadiazine IGR that disrupts molting by preventing chitin development (Uchida et al. 1985). It is active primarily against sucking insects such as scales, whiteflies, mealybugs and leafhoppers, although it also has activity against beetles. Buprofezin is slow-acting but persists a long time. It has poor ovicidal activity, but treated adults of some pest species may lay sterile eggs. It has little or no effect on lepidopteran (moths), dipteran (flies) or hymenopteran (wasp) insects. Buprofezin is toxic to crustaceans, limiting its use around bodies of water.
In citrus, buprofezin is not as effective as pyriproxyfen in controlling California red scale because it requires the majority of the scale population to be in an immature stage (Grout and Richards 1991). Buprofezin is less toxic than OPs and carbamates to a number of natural enemies, especially wasp parasites, allowing their numbers to greatly increase. However, similar to pyriproxyfen, it is highly toxic to coccinellid beetles, preventing larval molting (Grafton-Cardwell and Gu 2003). Buprofezin use has led to outbreaks of cottony cushion scale in citrus due to the loss of vedalia beetle. It also may be responsible for pest resurgences in other crops due to its effect on coccinellid predators.
Currently, buprofezin is used in California to control pests in almonds, citrus, cotton and grapes. Buprofezin has been an important component of the silverleaf whitefly management program for cotton, where it has helped delay resistance to insecticides. In grapes, buprofezin can be rotated with the neonicotinoids to control mealybugs and leafhoppers and so help to manage resistance. Because of its selectivity favoring hymenopteran parasites, buprofezin and parasites work together to control grape and vine mealybugs. Buprofezin is likely to receive registration for San Jose scale in stone fruit in the near future.
Pymetrozine
Pymetrozine is a pyridine azomethine. It is active primarily against sucking insects such as aphids and whiteflies. Its mode of action is not fully understood, but differs from other insecticide groups. It interferes with feeding behavior, resulting in the complete cessation of feeding within hours of contact (Harrewijn 1997). Aphids remain alive for 2 to 4 days before they die of starvation. Pymetrozine has been shown to reduce both direct damage and virus transmission by aphids (Bedford et al. 1998). Because of its specificity for sucking insects, it is relatively nontoxic to most natural enemies (Sechser et al. 2002).
Pymetrozine is active against important pests in cole crops, lettuce, celery and tomatoes. It has been registered for California cotton since 2001 to control cotton aphid (Aphis gossypii), but has not yet been incorporated into management programs to any extent. Pymetrozine must be applied when populations of cotton aphids are low, but in California the treatment threshold for aphid populations has traditionally been fairly high. When used for whitefly control, pymetrozine causes adults to stop feeding, but it must be used in combination with other insecticides such as IGRs to reduce whitefly populations below the economic threshold.
Controlling dipteran leafminers
Cyromazine (Trigard) is a triazine insecticide used as a chitin-synthesis-inhibitor IGR, which disrupts the molting of larval and pupal cuticles. It has translaminar activity that quickly penetrates into leaves. It is active against a very narrow range of insect pests, notably the larval stages of dipteran leafminers. Because of its high level of specificity, cyromazine is much less toxic to natural enemies, compared with IGRs such as diflubenzuron, making it highly compatible with integrated pest management (IPM) programs (Schuster 1994). Cyromazine is nontoxic to crustaceans, bees, fish and birds.
The current uses of cyromazine are for Liriomyza leafminers in cole crops, lettuce, peppers, spinach, celery, tomatoes and cucurbits (table 3). Cyromazine is important as a new chemistry for controlling dipteran leafminers, as a number of species have developed resistance to the older groups of insecticides around the world. Because leafminers often require multiple insecticide treatments, it will be important to rotate cyromazine with other insecticide chemistries to combat resistance.
A product with broad activity
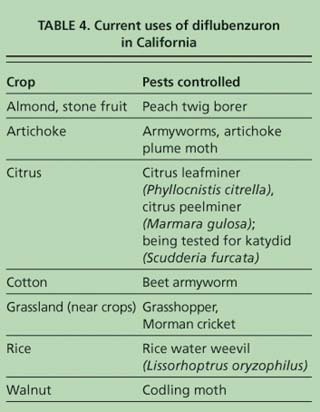
Diflubenzuron (Dimilin, Micromite) is a benzoylphenylurea chitin-synthesis-inhibitor IGR that disrupts molting. It is slow-acting, requiring up to 14 days for population reduction, because it is active against all molting stages. The symptoms of diflubenzuron poisoning in grasshoppers include slowed movement, uncoordinated jumping, loss of legs, decreased feeding and malformed wings (Weiland et al. 2002). It is used against a variety of insects including rice water weevils (Lissorhoptrus oryzophilus), beetles, various Lepidoptera, grasshoppers, Mormon crickets (Anabrus simplex) and katydids. It does not cause mortality to adults, but does sterilize the females of some species.
Because diflubenzuron is primarily active through ingestion, it is less toxic to a number of natural enemies, especially wasp parasites. Similar to other IGRs, the eggs and immature stages of predatory beetles, as well as lacewings, can be sensitive to diflubenzuron (Ables et al. 1977). However, effects such as reduced egg hatch can be rapidly reversed when the predators enter an untreated environment (Peleg 1983). Diflubenzuron is toxic to crustaceans; however, populations recover rapidly because it rapidly dissipates in water.
Currently, diflubenzuron is used to control pests in artichokes, citrus, cotton, rice, nuts and stone fruits (table 4). Diflubenzuron is also registered to treat grasslands infested with grasshoppers — which often devastate nearby crops — providing an important replacement for OP and carbamate insecticides. Resistance to diflubenzuron in codling moth populations occurs in France (Sauphanor et al. 2000), indicating the need for the careful rotation of this insecticide with insecticides that have different modes of action.
Pros and cons
Novel insecticide classes play a critical role in the IPM of many California crops. Excellent efficacy, high selectivity and low mammalian toxicity make them attractive replacements for OPs and carbamates, and the majority are considered by the U.S. Environmental Protection Agency to be “reduced risk” insecticides. However, their high level of selectivity can increase the need for other insecticides if they allow secondary pests that lack effective natural enemies to gain primary pest status. In addition, the new insecticides are not always completely selective, and the predatory beetles have been especially sensitive to some of them. Other problems include short residuality and high cost.
Nonetheless, major impetuses for the adoption of these chemistries include human health concerns and pest resistance to OPs, carbamates and pyrethroids. The wide variety of new modes of action is extremely helpful for delaying resistance in key pests such as whiteflies, scales and aphids. As growers and pest control advisors become familiar with the unique characteristics of these insecticides, their adoption is likely to increase.