All Issues
Phytoplankton fuels Delta food web
Publication Information
California Agriculture 57(4):104-109. https://doi.org/10.3733/ca.v057n04p104
Published October 01, 2003
PDF | Citation | Permissions
Abstract
Populations of certain fishes and invertebrates in the Sacramento-San Joaquin Delta have declined in abundance in recent decades and there is evidence that food supply is partly responsible. While many sources of organic matter in the Delta could be supporting fish populations indirectly through the food web (including aquatic vegetation and decaying organic matter from agricultural drainage), a careful accounting shows that phytoplankton is the dominant food source. Phytoplankton, communities of microscopic free-floating algae, are the most important food source on a Delta-wide scale when both food quantity and quality are taken into account. These microscopic algae have declined since the late 1960s. Fertilizer and pesticide runoff do not appear to be playing a direct role in long-term phytoplankton changes; rather, species invasions, increasing water transparency and fluctuations in water transport are responsible. Although the potential toxicity of herbicides and pesticides to plankton in the Delta is well documented, the ecological significance remains speculative. Nutrient inputs from agricultural runoff at current levels, in combination with increasing transparency, could result in harmful algal blooms.
Full text
William Sobzcak, former U.S. Geological Survey postdoctoral researcher, samples for zooplankton in the Delta.
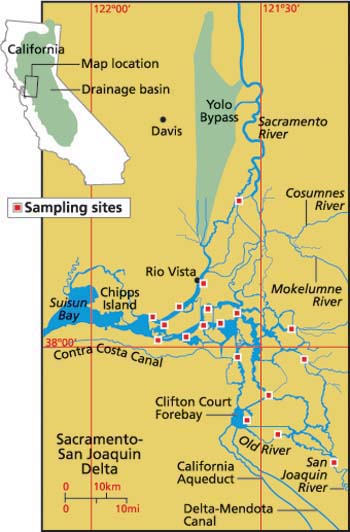
The Sacramento-San Joaquin River Delta is a complex mosaic of waterways that forms the transition zone between San Francisco Bay and its watershed (fig. 1). Over the last century, the original dominant marsh habitat has been lost through filling and diking. Water flows have changed radically. Exotic plants and animals have invaded or been introduced intentionally, and toxic contaminants have become widespread (CALFED 2000). The Delta is now a focus of ecosystem restoration because these changes have been accompanied by declines in the abundance of many fish species that use the Delta as a migration route, nursery or permanent habitat. Some species (thicktail chub) have already become extinct; others (winter-run chinook salmon) are now at risk of extinction; and still others (splittail, striped bass) have dramatically reduced populations.
Several lines of evidence suggest that food limitation has played a role in these declines. Many fish populations in the Delta are declining because of poor survival during the first year of life, which can be caused by food shortages (Bennett and Moyle 1996). Sport fish such as striped bass (Morone saxatilis), native species such as delta smelt (Hypomesus transpacificus), and commercial species such as chinook salmon (Oncorhynchus tshawytscha) all show evidence of food limitation during their first year. Zooplankton, a key food for young fishes, has also declined (Orsi and Mecum 1996). Some declining zooplankters, especially a mysid shrimp (Neomysis mercedis) and smaller species, also appear to be limited by food supply, as does the clam Corbicula fluminea, a dominant benthic invertebrate.
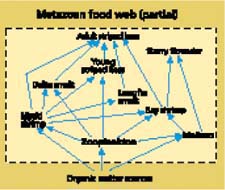
Zooplankton, benthic invertebrates and the larger, more visible fish and waterfowl that feed on them form a food web that depends ultimately on inputs of organic matter at its base (fig. 2). In many systems, phytoplankton species play a fundamental role in the organic matter supply to food webs. These microscopic plants are responsible for primary production, the photo-synthetic production of organic matter. Other sources of organic matter are often present, however, and can even be dominant, especially in estuaries. In fact, there has been speculation for many years that organic matter carried in from upstream and from adjacent terrestrial sources is the main source sustaining the Delta's food web. What, then, are the relative roles played by phytoplankton and other sources in the organic matter supply to Delta waterways? The question is a basic one for restoration of the Delta, because it determines the focus for increasing food supply to declining populations. In addition to investigating this, we examine evidence for a long-term decline in the primary food supply and consider the role of agriculture.
Food comes in many forms
To determine the most important organic matter sources for Delta waterways, we combined decades of data collected by the California Department of Water Resources (CDWR), U.S. Bureau of Reclamation (USBR) and U.S. Geological Survey (USGS), using a variety of estimation techniques (Jassby and Cloern 2000).
Glossary
Glossary
Algae:: Primitive plants usually living in water and occurring as single cells, filaments, colonies and irregular aggregations. Aquatic vascular plant: Higher plants living in water bodies. May be free-floating or rooted. Also known as aquatic macrophytes. Benthic invertebrates: Invertebrates living on or in the bottom sediments.
Benthic microalgae:: Microscopic algae living on or just under the surface of the bottom sediments.
Biomass:: The weight of biological matter, usually measured in terms of carbon, dry weight or fresh weight, and expressed on an areal (g m-2) or volumetric (mg m-3) basis.
Chlorophyll:: A green pigment present in most plants and essential for the process of photosynthesis, by which these plants obtain most if not all of their energy. Chlorophyll a is one form that is often assumed an approximate index of algal biomass.
Detritus:: Nonliving particulate and dissolved organic matter.
Metazoa:: Multicellular animals, in contrast to the more primitive protozoa.
Phytoplankton:: The plant plankton, consisting mostly of microscopic algae.
Plankton:: The community of passively suspended or only weakly swimming organisms in a body of water, which drift along with the water currents; planktonic organisms range in size from tiny plants and animals to large jellyfish, and include the larval stages of many fishes.
Primary productivity:: The rate at which plants incorporate inorganic carbon into organic matter. Usually measured on a volumetric (mg C m-3 d-1) or areal (mg C m-2 yr-1) basis. Primary production is often used to refer separately to the amount of organic carbon produced in a particular time interval (mg C m-3 or mg C m-2).
Protozoa:: Single-celled animals, including amoeba, ciliates and flagellates in aquatic systems.
Suspended sediments:: Small mineral (clay and silt) particles suspended in waters.
Turbidity:: The scattering effect that suspended and dissolved solids have on light, imparting a cloudy appearance to water. Primary contributors include suspended sediments, soluble colored organic compounds and microscopic organisms.
Zooplankton:: The animal plankton, in estuaries consisting mostly of protozoa, rotifers and two crustacean types, the cladocerans and the copepods.
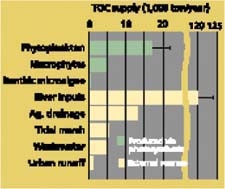
Organic matter sources are usually compared in terms of their total organic carbon (TOC) content; TOC can come in both particulate (POC) and dissolved (DOC) forms. River input — organic material carried in by the Sacramento, San Joaquin and other rivers and creeks — is the largest TOC source overall (fig. 3). Some of this river-borne material is actually phytoplankton and phytoplankton-derived detritus transported from upstream of the Delta.
Phytoplankton production within the Delta and organic matter in agricultural drainage directly into the Delta are next in importance. Agricultural drainage upstream of the Delta is included in river inputs and cannot be estimated separately with any confidence. Much of the organic matter in agricultural drainage originates from leaching of DOC from island peat soils. Discharge from wastewater treatment plants, drainage from tidal marshes and production of aquatic vascular plants such as the submerged Egeria densa are tertiary sources. Urban runoff, primary production by benthic microalgae and other sources — although sometimes important in other estuaries — are negligible sources of organic matter in the Delta's Phytoplankton is clearly the Delta's dominant primary producer, whereas river input of organic material from upstream is dominant among external sources (fig. 3). Many of these sources are distributed unevenly throughout the Delta, and we can identify areas where aquatic vascular plants, tidal marsh drainage or another source may dominate at times. Nevertheless, phytoplankton production, river inputs and agricultural drainage together account for 90% of average annual Delta-wide organic matter sources.
Fig. 1. The Sacramento-San Joaquin River Delta extends upstream from Chipps Island to include leveed islands, river channels, sloughs, flooded islands and tidal marshes. Boxes mark stations used by the California Department of Water Resources to assess water quality, in some cases since the late 1960s.
Fig. 2. The metazoan food web can be extremely complex, especially in estuaries, and only a small portion of the Delta's food web is portrayed here. The energy and nutrient needs of the metazoan food web are supported by a variety of organic matter sources.
Fig. 3. Organic matter, measured as total organic carbon (TOC), is produced in Delta waterways by photosynthesis and enters from a variety of external sources. Phytoplankton is the biggest internal source and river transport is the biggest external source. Error bars represent standard error among years.
Drinking water and organic matter
Aside from its ecological significance, organic matter also has implications for drinking-water quality in the Delta. The Delta provides all or part of the drinking-water supply for about 22 million California residents. When disinfectants such as chlorine are added to drinking water to kill microbial pathogens, they react with bromide and naturally occurring organic matter to form disinfection byproducts (DBPs). The main DBP groups are total trihalomethanes, haloacetic acids, bromate and chlorite. When these are consumed over years in excess of federal standards, some people may experience problems with the liver, kidneys or central nervous system, or may have an increased risk of cancer or anemia (Bull and Kopfler 1991).
A current major challenge for water suppliers in the Delta and elsewhere is how to balance the risks from pathogens and DBPs; it is important to provide protection from these pathogens by using disinfectants while simultaneously containing health risks from DBPs. One way to limit DBP formation is to limit TOC levels in raw water supplies. Our organic matter assessment implies that phytoplankton production, river-borne loading and agricultural drainage should each be a focus of source-control measures with respect to the DBP problem: they are all important sources of naturally occurring organic matter in the Delta.
The participation of phytoplankton in ecosystem food supply and drinking-water quality points to one example of conflicting aims in the Delta. Although higher phytoplankton production may be a boon to certain food-limited organisms, it can degrade drinking-water quality through the formation of DBPs. The diversity of issues in the Delta creates a complex balancing problem: human health versus ecosystem health. The balancing of different aims is particularly difficult with regard to phytoplankton, which has many other effects. Negative impacts include clogging filters, producing undesirable tastes and odors, and contributing dangerous substances directly to raw water, such as the liver toxin microcystin-LR and neurotoxin anatoxin. On the positive side, phytoplankton is central in the bioconcentration of contaminants, transport and cycling of plant nutrients, and the atmospheric carbon dioxide balance.
An inefficient food source
The bulk accounting in figure 3 is an inadequate guide to the relative value of different organic materials for primary consumers such as zooplankton and clams. Particulate and dissolved forms of organic matter differ markedly in their availability to the food web, making further refinement necessary. POC enters the Delta mostly as phytoplankton, bacteria and protozoa, particles of decaying organic matter, and suspended mineral particles carrying organic matter on their surfaces. These particles can be utilized directly by primary consumers such as clams and zooplankton.
In contrast, most dissolved organic carbon must first be transformed into particles before it can be consumed. This transformation happens primarily via the uptake and metabolism of DOC by bacteria; in other words, the DOC is converted to bacteria cells. Much of the DOC is not very bioavailable (not easily assimilated and metabolized) and is simply flushed downstream before bacteria can utilize it. Moreover, much of the remaining DOC that is metabolized by bacteria is lost to respiration and does not end up as bacterial biomass.
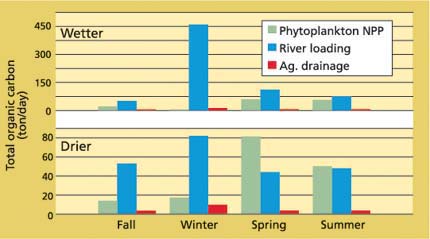
We refined our estimates of the three major organic-matter sources by accounting for bioavailability of DOC and respiratory losses, based on generalizations from previous empirical studies (Jassby and Cloern 2000). We then categorized each year as either drier or wetter than average and plotted the refined estimates of TOC in each category by season (fig. 4). The relative importance of organic matter sources changes dramatically because so much of river input and agricultural drainage is in dissolved form. Phytoplankton production is seen as a significant source of bioavailable organic matter in all seasons, except for winters with above-normal precipitation. Moreover, phytoplankton production is comparable to and sometimes greater than river inputs in spring and summer of both above-normal and below-normal years. Spring and summer are particularly critical seasons for survival and growth of young fish and successful recruitment to fish populations. In contrast, agricultural drainage is almost never a significant source of bioavailable organic matter. Consequently, plans to control organic matter in agricultural and Delta island drainage because of the DBP problem should have little impact on food supply to the Delta's food web.
Recently this work was corroborated with an extensive set of bioassays that assessed the food value of the Delta's organic matter sources (Sobczak et al. 2002). Although dissolved organic matter is the major energy and carbon source for bacterial metabolism, the dominant food supply to the planktonic food web is bioavailable, particulate organic matter derived primarily from internal phytoplankton production.
Differing nutritional quality
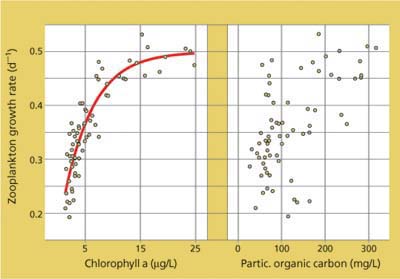
Just as dissolved organic-matter sources contribute little to the food supply compared with particulate sources, the particulate sources themselves vary in quality. Delta phytoplankton are a better food source for zooplankton than other kinds of particles in the POC pool — decaying organic matter, bacteria and organic matter clinging to the surface of clay and silt particles. The zooplankter Daphnia magna, which occurs in the Delta, feeds nonselectively on particles smaller than 40 micrometers (μm). In a series of laboratory feeding assays (Müller-Solger et al. 2002), Daphnia were exposed using a flow-through system to water from four Delta habitat types collected during all four seasons, and growth rates were measured. While POC concentrations were only weakly related to Daphnia growth, concentrations up to a threshold of about 10 μg/L chlorophyll a — a pigment found in phytoplankton — predicted Daphnia growth rates across all habitats and seasons (fig. 5). Chlorophyll is not a nutrient; it is merely a marker for the phytoplankton fraction of particulate organic matter and a convenient way to estimate phytoplankton biomass. The actual nutritional factors in phytoplankton determining Daphnia growth rates are not known for certain, although elements such as phosphorus and certain essential fatty acids are candidates.
Fig. 4. Phytoplankton net primary productivity (NPP), river loading and agricultural drainage make up most of the bulk organic-matter supply. The values shown here have been corrected for losses due to lack of bioavailability and respiration, and are therefore a more realistic comparison of the food value for consumer organisms than the bulk data of figure 3. Phytoplankton provides a significant source especially in spring and summer, a critical period for populations of many fishes and invertebrates.
Fig. 5. The growth of the zooplankter Daphnia magna in Delta waters is closely related to the supply of phytoplankton, as indexed by chlorophyll a concentrations, but not so closely tied to levels of particulate organic carbon in general.
Supply changes year to year
Phytoplankton are probably the most important portion of the particulate organic matter supply to the Delta's food web on a Delta-wide basis, and growth rates of primary consumers such as Daphnia are closely tied to phytoplankton availability below about 10 μg/L. But how often is Delta phytoplankton at levels that can limit zooplankton growth, and is phytoplankton variable enough to induce major changes in zooplankton growth rates? In fact, Delta chlorophyll levels quite commonly fall within the range limiting growth. Thousands of chlorophyll a measurements have been made in the Delta since the late 1960s by the CDWR and USBR, and 55% to 93% of them, depending on the year, are below 10 μ/L (Jassby et al. 2002). Moreover, large swings have occurred in Delta-wide chlorophyll from one year to the next, and longer-term changes are evident.
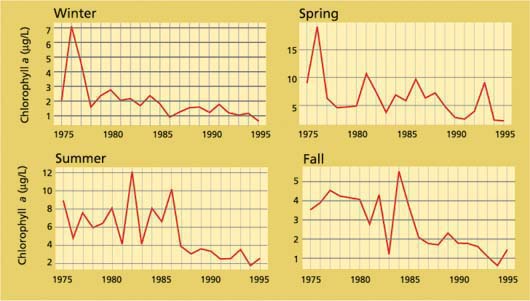
Figure 6 shows the annual average of Delta-wide chlorophyll for each season during a period when analytical methods remained the same and sampling was sufficiently comprehensive to cover the entire Delta. Although the trend is not uniformly downward, there has been an overall tendency toward lower phytoplankton concentrations in later years. In fact, all except spring months (April to June) showed a robust, statistically significant downward movement from 1975 to 1995 (Jassby et al. 2002). Phytoplankton variability could therefore easily lead to a several-fold difference in zooplankton growth rates.
What are the reasons for this variability? In 1986, an Asian clam (Potamocor-bula amurensis) invaded and established itself in Suisun Bay, presumably after being discharged with ship ballast water. Its establishment and dispersal were aided by the prolonged drought and accompanying low freshwater inflows to the Delta that began in 1987. Potamocorbula turned out to be a voracious consumer of phytoplankton and changed phytoplankton dynamics in Suisun Bay (Alpine and Cloern 1992). The effects of Potamocorbula probably extend into the western Delta, with a summer downturn after 1986 even in the Delta-wide chlorophyll record (fig. 6); Potamocorbula are feeding most actively during summer.
A recent analysis identified the most important driving forces for Delta-wide phytoplankton production: interannual variability of water flow; increased consumption by Potamocorbula; and a downward trend in suspended mineral particles over many decades, which improves water transparency and therefore phytoplankton photosynthesis and growth rate (Jassby et al. 2002). The increase in phytoplankton growth rate partially compensates for increased losses due to consumption by clams, but apparently not by enough to prevent a decrease in phytoplankton biomass. The dry weight of suspended mineral particles is much greater than phytoplankton biomass: variations in the latter have relatively little effect on transparency.
Impact of agricultural runoff
Dissolved organic matter in agricultural drainage, although an important issue for drinking-water quality, is not a significant source of energy for the Delta's food web. What effects might other constituents of drainage and runoff, namely pesticides and nutrients, have on phytoplankton productivity?
Pesticide toxicity.
Herbicide concentrations may limit phytoplankton growth rates during localized occurrences of elevated concentrations. In 1997, Jody Edmunds and colleagues at the USGS examined 53 water samples collected from May through September at nine Delta sites for six herbicides that inhibit photosynthesis. Only one sample exceeded concentrations (diuron) reported to inhibit primary production in laboratory experiments (Edmunds et al. 1999). Similarly, bioassays showed no relationship between ambient herbicide concentrations and photosynthesis, except for this one sample. The study design might have missed herbicide runoff events during the rainy season.
In fact, Jeff Miller and others from the Central Valley Regional Water Quality Control Board, using an algal indicator species, found toxicity in 22% of samples from the Sacramento-San Joaquin watershed and Delta during 2000 to 2001 (Miller et al. 2002). Again, diuron — an herbicide applied to rights-of-way, alfalfa, vineyards and orchards — was implicated. Most toxicity occurred from January to March when diuron is applied and when it is most likely to rain in California; in contrast, most phytoplankton production takes place in spring and summer. The ecological consequences of this photosynthetic inhibition may therefore be limited.
Fig. 6. Average chlorophyll a, and therefore phytoplankton biomass, is highly variable from season to season and year to year in the Delta, but in general it has been decreasing since at least the 1970s. Summer chlorophyll, in particular, decreased markedly after the Asian clam, Potamocorbula amurensis, invaded in 1986; the clams feed most actively during summer.
Pesticides may also affect primary production through indirect effects on the zooplankton community, molluscs and other organisms that feed on phytoplankton. Data collected by the USGS National Water-Quality Assessment program demonstrate that seven pesticides in the San Joaquin River Basin frequently exceed criteria for the protection of aquatic life, and diazinon concentrations sometimes reach acutely toxic levels in the San Joaquin River (Dubrovsky et al. 1998). Similarly, diazinon and chlorpyrifos are linked to toxicity in test zooplankton in the Sacramento River watershed. The Regional Monitoring Program of the San Francisco Estuary Institute has established that these organophosphate pesticides are also of concern in San Francisco Bay. While there is a growing body of information that pesticides in surface-water runoff can be toxic to invertebrates in the San Francisco Bay and Delta, the ecological significance has not yet been established.
Nutrient levels.
Largely because of agricultural drainage, nutrient supplies are well in excess of phytoplankton needs in the Delta. The availability of nitrogen and phosphorus are important determinants of phytoplankton growth and biomass in many aquatic systems. A low nutrient supply can restrict the growth of phytoplankton and, ultimately, fish yield. We found that nutrient concentrations were low enough to limit phytoplankton growth for only about 0.1% of the measurements since the late 1960s, most occurring in the southern Delta during the extremely dry El Niño-Southern Oscillation of 1976 to 1977 (Jassby et al. 2002). Nutrient sources and this nutrient excess are not as pronounced downstream in San Francisco Bay, and nitrogen can become limiting during spring phytoplankton blooms in the South Bay.
Excessive nutrients from agricultural drainage or animal wastewater have promoted huge and harmful phytoplankton blooms in many locations around the world (Anderson et al. 2002). This is not a major problem in the Delta currently because of high concentrations of suspended sediments and accompanying turbidity. High turbidity is in part a legacy of the erosion caused by hydraulic mining in the Sierra Nevada in the 19th century. By decreasing transparency and limiting the penetration of sunlight, turbidity slows phytoplankton photosynthesis and limits its ability to reproduce rapidly to massive levels. However, suspended sediment in the Delta has been decreasing and transparency increasing for decades.
USGS scientists have identified several possible explanations for this phenomenon (Wright and Schoellhamer in press). First, reservoirs have been trapping sediment behind dams similar to the decrease in sediment load over the past 50 years. Second, there are still channel and floodplain deposits of mining-derived sediments that are being eroded and gradually depleted. Third, bank stabilization such as riprap retards meandering, eliminating a sediment source (channel banks) and contributing to decreasing sediment yield. Finally, the depositional nature of the lower Sacramento floodplain has changed in a way that, in principle, could trap additional sediment. The relative importance of these mechanisms is not known precisely. In any case, given the excess of nutrients in the Delta, decreasing turbidity means that large phytoplankton blooms may become a more common phenomenon (Jassby et al. 2002). Moreover, Delta waters are warming, and higher temperatures favor the cyanobacteria (blue-green algae) that constitute nuisance or harmful algal blooms. If such nuisance or harmful blooms become common, control of nitrogen and phosphorus inputs from agricultural drainage will become a much more important issue.
Ecosystem restoration
The research described here highlights the importance of phytoplankton in sustaining the metazoan food web on a Delta-wide basis, despite the presence of many other organic matter sources. Organic matter in agricultural drainage is mostly in dissolved form and not an important nutrient or energy source for the metazoan food web; along with phytoplankton and other sources, however, it reacts with disinfectants during drinking-water treatment to form potentially harmful byproducts. Phytoplankton biomass — and therefore the food supply for higher organisms — has declined over the past few decades, partly because of the Asian clam invasion. Certain herbicides in agricultural drainage may at times inhibit phytoplankton production, especially in winter, but their overall effect on annual production is probably limited. Similarly, certain pesticides can reach toxic levels for primary consumers of phytoplankton, but any ecological significance has not yet been demonstrated.
The phytoplankton decline may represent a reduction in the system's capacity to support higher levels of the food web. Lower phytoplankton levels have been linked to declines in key zooplankton populations in the Delta. Although the evidence for food limitation of fish populations is not as strong as for zoo-plankton and benthic invertebrates, data from many estuaries and other water bodies also points to an overall correspondence between fish production and primary production (Nixon and Buckley 2002). Unless phytoplankton productivity increases, restoration of fish populations in the Bay-Delta may be limited. Water transparency has increased over the past few decades due to declines in suspended sediments, enhancing phytoplankton photosynthesis and partially offsetting consumption by clams. A continuation of the transparency trend could result in increased phytoplankton production because of the excess nutrients available in the estuary from fertilizer runoff and wastewater treatment effluent. Although this could have positive effects on overall fish production, there is a potential danger from nuisance and harmful phytoplankton species that pose both human and ecological toxicity problems.
As a response to symptoms of gross ecosystem disturbance and the critical role of the Delta as the linkage between San Francisco Bay and its watershed, a consortium of state and federal agencies was established in 1994. The CALFED Bay-Delta Program's mission is to develop a long-term and comprehensive plan to “restore ecological health and improve water management for the beneficial uses of the Bay-Delta system.” The program is centered around four objectives, one of which focuses on environmental quality to “improve and increase aquatic and terrestrial habitats and improve ecological functions in the Bay-Delta to support sustainable populations of diverse and valuable plant and animal species” (CALFED 2000). This is one of the largest attempts at ecosystem restoration worldwide, with a multibillion dollar budget and a period of 25 to 30 years for full implementation. Addressing the decline in system productivity is part of one of the key strategic goals of this ecosystem restoration.
Attainment of the CALFED Bay-Delta Program restoration goals requires a solid base of scientific understanding to identify key ecosystem functions within the Delta and to describe how they change in response to human activities, including restoration actions. Restoration actions — including new canals, flow and fish barriers, increased use of floodplains and increased shallow-water habitat — all have significant impacts on phyto-plankton production, some positive and some negative (Jassby and Cloern 2000). Given the significance of phyto-plankton production to the food base in the Delta, as well as other phytoplankton-related functions, these impacts must be defined quantitatively and used to help guide the restoration strategy.