All Issues
Grape growers report losses to black-foot and grapevine decline
Publication Information
California Agriculture 52(4):19-23. https://doi.org/10.3733/ca.v052n04p19
Published July 01, 1998
PDF | Citation | Permissions
Abstract
Sporadic but increasing incidence of two new diseases, which we have named “black-foot disease” and “Phaeoacremonium grapevine decline,” are taking a toll in California's table and wine grape vineyards (Scheck et al 1998a, 1998b). Although the full scope of the problem is not yet measured, grape growers throughout major production regions in California have reported economic losses from replanting costs. Symptoms of these diseases are almost indistinguishable from each other; the diseases are in fact the same that have occurred in grapevines in other places in the world and in older grapevines here. Our research reveals that black-foot is caused by Cylindrocarpon obtusisporum, and grapevine decline is caused by Phaeoacremonium species (P. aleophilum, P. chlamydosporum, or P. inflatipes).
Full text
Starting in the early 1990s, growers throughout California began reporting decline symptoms in young grapevines. Decline was particularly evident in areas where growers had extensively replanted phylloxera-resistant rootstocks (see California Agriculture, July-August 1996). Although incidents were isolated, they steadily increased in number, frequency and distribution throughout major production regions, often forcing growers to replant young vineyards that had established slowly and shown poor early growth.
The field symptoms of black-foot disease and young grapevine decline are nearly indistinguishable, and are referred to here collectively as “decline symptoms.” In tests of young grapevines with decline symptoms, we isolated fungi from the genus Cylindrocarpon, which we determined caused “black-foot disease,” and from the genus Phaeoacremonium, which we determined caused “Phaeoacremonium grapevine decline”(also known as “young grapevine decline.”) While neither disease poses a devastating threat to the grape industry of California, replanting costs can be significant. Many growers opt to replant only the weakest vines, but some are replanting sizable acreages where young grapevines have been growing very slowly.
Above, foliage symptoms of Phaeoacremonium young grapevine decline (Vitis vinifera ‘Thompson seedless’).
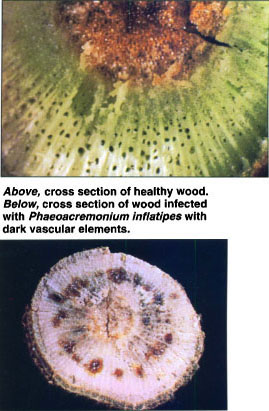
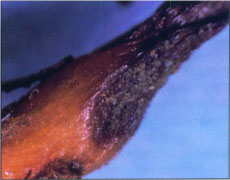
Above, cross section of healthy wood. Below, cross section of wood infected with Phaeoacremonium inflatipes with dark vascular elements.
Editor's note
With this issue, California Agriculture introduces “New pests and plant diseases,” survey articles describing new pests and diseases of statewide significance. We are publishing this new research section as an experiment. In the last 3 years, California Agriculture has not printed articles of this nature because they do not meet the test of containing 2 years of experimental data. However, we feel that these articles may provide Important information to our readers. Please let us know what you think about this, and whether articles of this nature present Information Important to you. Ed.
Little is known about the etiology or epidemiology of these pathogens on young grapevines. There is a link between young grapevine decline and “black measles,” one of the most destructive diseases of woody tissues in older grapevines. The same Phaeoacremonium spp. have been implicated as the pathogens responsible for both diseases. Although black measles has been observed in California for 50 years, it has proven difficult to characterize, partly due to its variability in symptom expression between years. In addition, until now that disease has only affected older grapevines. We do not know why pathogens of older vines would emerge as a threat to young vines, but older vines carrying black measles may be one source of the new fungal diseases.
The pathogen for black-foot disease is better understood because it has a wider host range, but the reasons for its increasing incidence are not well understood. Because of these two new threats to young vines, we emphasize the importance of cultural techniques to minimize stresses, both preplanting and postplanting. For these, growers should contact their local farm advisors.
Symptoms
The diseases produce overlapping symptoms and generally follow a similar pattern of development. Growth of affected grapevines slows and differences in vigor are typified by reduced caliper size of the trunk, shortened internodes, reduced foliage, and reduced leaf size. Grapevines that appear healthy in cross section at planting begin their first season with adequate vegetative vigor and development. However, during the subsequent 3 to 5 years after planting, the onset of foliar symptoms may appear as interveinal chlorosis, followed by necrosis, and early defoliation. When trunks of declining grapevines are viewed in cross section, dark-brown to black streaking in the vascular elements is evident. This discoloration may occur in a few to most of the vascular elements. Uneven wood maturity, which is usually associated with a rapid desiccation event, is another telltale symptom\. Below ground, symptoms include a reduction in total root biomass, low numbers of feeder roots, and sunken, necrotic root lesions.
Isolation results
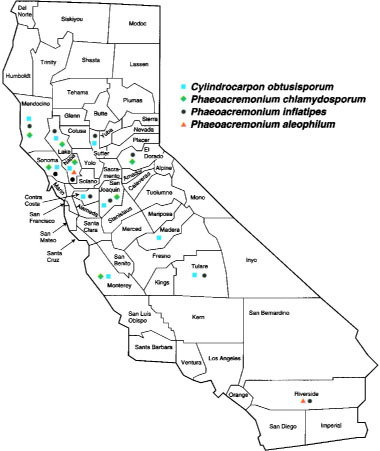
Symptomatic grapevines were submitted by UCCE farm advisors, pest control advisors and vineyard managers. Additionally, many collection trips were made to observe affected vineyards. We have consistently isolated four species of plant pathogenic fungi from symptomatic specimens of young grapevines from Northern, Central, and Southern California counties (fig. 1).
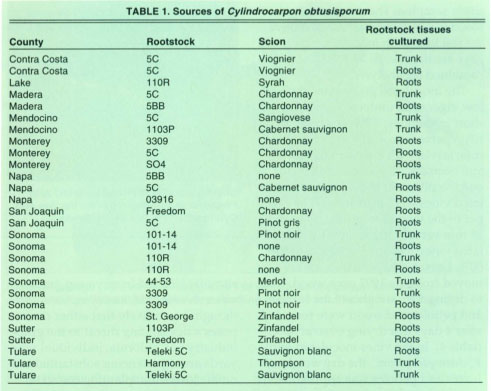
Cylindrocarpon obtusisporum was isolated from the roots and trunks of 2- to 5-year-old declining grapevines in Contra Costa, Lake, Madera, Mendocino, Monterey, Napa, San Joaquin, Sonoma, Sutter and Tulare counties (table 1). Root isolations were made from the margins of necrotic brown-colored and healthy tan-colored root tissues. Isolation attempts from the rootstock trunks were from discolored areas of the pith and vascular tissue. C. obtusisporum grew rapidly from the infected tissue pieces and sporulated readily, producing abundant microconidia, macroconidia, and chlamydospores in culture.
Cylindrocarpon destructans, a pathogenic species closely related to C. obtusisporum, was first reported in France in 1961 as the cause of black-foot disease on young vines. A recent study of one vineyard in the Bordeaux region of France found that up to 50% of grapevine mortality occurred in new plantings because of this pathogen (Maluta and Larignon 1991). Cylindrocarpon spp. are soil-borne and infect grapevines through natural openings or wounds on the roots or other belowground portions of the rootstock such as the pith. Over time the fungus invades lignified tissues of the plant, resulting in a brown or black necrosis of the roots and vascular elements.
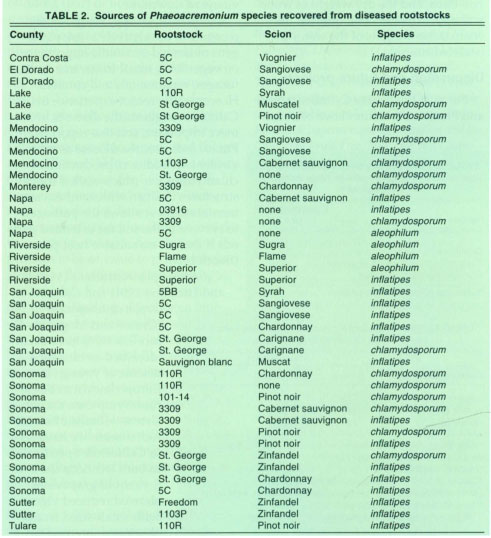
Grapevine diseases associated with Phaeoacremonium spp. include black measles (or “esca”), and apoplexy, and have been observed in Europe and South Africa (Crous et. al. 1996; Larignon and Dubos 1997). Three species of Phaeoacremonium (formerly Cephalosporium = Acremonium or Phialophora) were isolated from declining young grapevines in Northern, Central and Southern California from 1995 to 1997. Rootstock isolations were made from the pith and woody tissue of belowground portions of the root-stock. Diseased pith tissue was dark in color and wood, outside the pith, was dry with a silvery sheen. Phaeoacremonium spp. were found in the crown and lower portions of the rootstock of young grapevines with decline symptoms. These fungi grew slowly out of infected tissue, but sporulated abundantly in culture. The species Phaeoacremonium chlatnydosporum was isolated from wine grapes in El Dorado, Lake, Mendocino, Monterey, Napa, San Joaquin, and Sonoma counties; P. inflatipes was isolated from wine grapes in Contra Costa, El Dorado, Lake, Mendocino, Napa, San Joaquin, Sonoma and Sutter counties, and from table grapes in Riverside and Tulare counties; P. aleophilum was isolated from wine grapes in Napa County and from table grapes in Riverside County (table 2).
Pathogenicity experiments
Greenhouse experiments have demonstrated that Cylindrocarpon obtusisporum, Phaeoacremonium chlamydosporum, and P. inflatipes are aggressive pathogens of grape seedlings. One-month-old seedlings (Vitis vinifera ‘Carignane’) were inoculated by dipping their roots in a 106 spore/ml suspension for 30 minutes. After 60 days, 92% of seedlings inoculated with C. obtusisporum; 67% with P. chlamydosporum, and 71% with P. inflatipes had died (table 3). The pathogens were easily recovered from both the roots and the stems of the moribund seedlings. Control plants treated with water remained healthy and pathogens were never detected.
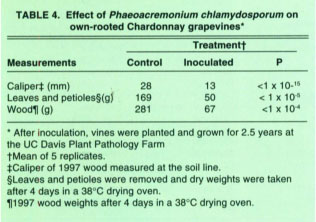
Field inoculations to confirm Phaeoacremonium as a pathogen of young vines were done at the Plant Pathology Farm, University of California, Davis. In August 1994, own-rooted grapevines (Vitis vinifera ‘Chardonnay’) were inoculated with a 106 spore/ml suspension which was introduced by drilling a small hole into the crown or by cutting the roots and dipping them into the spore suspension for 30 minutes and then planting in 3.8 liter pots. Control plants were treated in the same manner, but inoculated with sterile water. In February 1995, the plants were established in the field. In the winter of 1997, the grapevines were pruned to 2 buds. In October 1997, the test grapevines were removed from the field and destructively sampled. The caliper of the 1997 wood was measured at 2.5 cm above the soil line. Both inoculation methods gave similar results and the data were combined for analysis.
The inoculated grapevines showed low vigor with undersized trunks, short internodes, uneven wood maturity, sparse foliage and stunted, chlorotic leaves with interveinal chlorosis and necrosis. The vascular elements and the pith near the crowns of inoculated vines were discolored. The caliper of the wood for control vines was 28 mm versus only 13 mm for inoculated vines, a reduction of more than 50%. Leaves and petioles were removed from the 1997 cane wood prior to drying. Dry weights of the leaves and petioles and wood were recorded after 4 days in a drying oven at 38°C (table 4). In the vines inoculated with P. chlamydosporum, the dry weight of leaves and petioles was approximately one-third, and the dry weight of wood was approximately one-quarter, the comparable weights of the water-inoculated controls.
Uneven wood maturity associated with Phaeoacremonium grapevine decline and Cylindrocarpon black-foot disease.
TABLE 3. Pathogenicity tests with three fungi; inoculations of grape seedlings (Vitis vinifera L. ‘Carignane’)
Discussion and future projects
Fungi in the genera Cylindrocarpon and Phaeoacremonium have been consistently isolated from young grapevines showing decline symptoms. Although it is unlikely that either disease poses a devastating threat to the grape industry of California, individual vineyards are experiencing substantial economic losses in replanting costs and vineyard downtime.
Cylindrocarpon spp. are generally recognized as relatively weak pathogens of limited economic importance on vegetables, small fruits, tree fruits, forages, ornamentals and conifers. However, the recent experience in California suggests the diseases are more important, and one report from France indicates the disease as more virulent. In addition, production of chlamydospores (thick-walled fungal structures that can withstand environmental stresses) allows the pathogen to survive in the soil for extended periods if there is no suitable host plant (Booth 1966).
Cylindrocarpon destructans (Maluta and Larignon 1991) and Cylindrocarpon obtusisporum (Grasso and Magnano-disan-Lio 1975) have been identified as causing a disease of young vines in Europe known as black-foot. Symptoms are consistent with the black-foot disease we have seen in California. Specifically, in both locations the 2- to 5-year-old grapevines showed reduced vigor with small-sized trunks, shortened internodes, uneven wood maturity, sparse foliage and small leaves with interveinal chlorosis and necrosis. In cross section, trunks showed dark-brown to black streaking in a few to most of the vascular elements. Symptoms included a reduction in root biomass and root hairs with sunken, necrotic root lesions. Pith of affected vines was compacted and discolored. Unlike the situation in France where young grapevines were killed by Cylindrocarpon, mortality in California from black-foot disease has been low and infected vines struggle but continue to grow and develop, albeit poorly. One of the California isolates of Cylindrocarpon was identified as C. destructans in 1994. Since 1994, C. obtusisporum has been the most commonly isolated species. However, because of the similarities in symptom expression caused by these two species which are closely related to one another, we propose to maintain the name “black-foot disease” for those caused by either species of Cylindrocarpon.
We propose the name “Phaeoacremonium grapevine decline” to define the symptoms associated with infection by Phaeoacremonium spp. The same fungal species have also been implicated as the pathogens in black measles (or esca) disease of older grapevines (Crous et al, 1996; Larignon and Dubos 1997; Vasquez and Gubler, unpublished data). Black measles is one of the most destructive diseases of woody tissues in grape and has an extensive range of symptoms. These include stunted growth and die-back, plugged xylem vessels, leaf chlorosis and necrosis, raisining of fruit, and occasionally sudden wilting and/or death of vines. While black measles symptoms are very similar to those associated with Phaeoacremonium grapevine decline, it is unclear why pathogens formerly associated only with older grapevines have emerged recently as a threat to young grapevines. Perhaps cultural practices, such as poor irrigation management, or environmental conditions have shifted in the favor of the pathogens. Or perhaps new strains of these pathogens have evolved that are more aggressive toward young grapevines.
So far, no obvious relationship exists between grape rootstock/scion variety combinations and the severity of either Cylindrocarpon black-foot disease or Phaeoacremonium grapevine decline. Because of the phylloxera epidemic, the AXR rootstock, used almost exclusively for 20 years, has been replaced by other rootstock varieties. These may prove to be highly susceptible to these diseases, particularly if predisposed by stresses including insufficient irrigation of young vines or poor planting techniques. Investigations into differences in susceptibility and symptom expression in rootstock varieties are in progress.
Due to the similarity of symptoms, diagnosis of Cylindrocarpon black-foot disease or Phaeoacremonium grapevine decline should never be made solely on the basis of visual symptoms. (Growers should send samples to UC or California Department of Agriculture laboratories identification through isolation of the pathogen.) In addition, young grapevines can fail for a large number of reasons including poor nutrition and poor soil fertility, lack of adequate irrigation, poor planting techniques, virus-induced incompatibility, nematodes, and poor quality planting stock. Vascular symptoms can also have many biotic and abiotic causes. Black or amber streaks seen in the vascular elements are the result of deposition of phenolic compounds in response to wounding, a general defense mechanism of the plant. Physical injuries to the grapevine due to root tearing, disbudding (part of normal rootstock propagation), or other trauma during production or planting can result in phenolic depositions and black discoloration, or even outright death of tissue as the result of injury to cambium tissues. The popular press has described phenolic deposition and formation of gums and tyloses in the vascular tissues as “black goo” and “black xylem disease.” However, these names do not encompass the full range of symptoms associated with Cylindrocarpon black-foot disease or Phaeoacremonium grapevine decline, nor do they provide enough description of the problem to be of value to plant pathologists or to the viticulture industry.
Our lab is focusing on increasing our understanding of pathogen biology, disease epidemiology, and symptom development in Cylindrocarpon black-foot disease and Phaeoacremonium grapevine decline. Studies are underway to determine how these pathogens survive and spread through time and space. Sources of pathogen inoculum will be identified. With an increased understanding of the disease biology, effective control strategies will be developed.