All Issues
Aerial application of clopyralid demonstrates little drift potential and low toxicity to toads
Publication Information
California Agriculture 58(3):154-158. https://doi.org/10.3733/ca.v058n03p154
Published July 01, 2004
PDF | Citation | Permissions
Abstract
The herbicide clopyralid (Transline) is commonly applied by air to control yellow starthistle, a noxious weed, in California. In laboratory studies, clopyralid toxicity in Fowler's toad was low, indicating a wide safety margin when used under field conditions. In addition, monitoring of clopyralid drift following aerial application demonstrated that 98-foot (30-meter) buffers between treatment areas and water sources provided adequate drift protection for an adjacent stream and vernal pools. Nevertheless, to ensure that movement of the herbicide to water sources is minimized, it is important to prevent application error, particularly accidental encroachment into established buffer zones. This study demonstrated that drift potential for clopyralid was minimal even with an aerial application and a slight downwind breeze toward sensitive aquatic sites. It is also the first report demonstrating a high tolerance to clopyralid in larval toads.
Full text
Yellow starthistle is an annual, Eurasian weed commonly found along many of California's open roadsides, rangeland, wildlands, pastures and disturbed places (Maddox 1981). Since 1960, yellow starthistle has spread rapidly and currently infests an estimated 12 million to 20 million acres in California (DiTomaso, Kyser et al. 1999). Throughout the state, yellow starthistle (Centaurea solstitialis) decreases rangeland utilization, lowers forage quality and production, hinders military and recreational land use, and displaces native plant and animal habitats.
To control yellow starthistle, a helicopter applies clopyralid (Transline). This herbicide has a broad timing window and very low toxicity, with no grazing restrictions.
Several management methods can be used to control yellow starthistle including tillage, mowing, grazing, biological control, herbicides and revegetation with competitive perennial species (DiTomaso, Lanini et al. 1999; (DiTomaso et al. 2000; Thomsen et al. 1996). With the 1998 registration of clopyralid (Transline) for wildland use in California, aerial application has become a common, effective control method for yellow starthistle. Clopyralid is a picolinic acid herbicide that mimics the activity of naturally occurring auxins in plants; it is used at low rates and is selective for the control of some broadleaf species, particularly members of the sunflower and pea families. In addition, clopyralid has a broad timing window, does not appear to negatively impact insect biological-control agents (Pitcairn and DiTomaso 2000) and has a very low toxicity (signal word: “caution”) with no grazing restrictions.
Fort Hunter Liggett (FHL) is a 260-square-mile military installation located in southern Monterey County, on the eastern side of the Santa Lucia Mountains. The property is largely wildland and contains a number of habitats, including chaparral, woodland, grassland, vernal pools and marshes. A remarkable number of animal and plant species, several of them rare, are present. A state-listed species of concern, the California tiger salamander (Ambystoma californiense), and a federally listed endangered animal, the arroyo toad (Bufo californicus), are found on FHL. Yellow starthistle was first mapped on FHL in 1964, covering 1,660 acres. By 1998, it had spread to more than 20,000 acres (Osborne 1998). Yellow starthistle infestations are particularly dense in the areas along the San Antonio River and Nacimiento River riparian corridors, prime habitat for the arroyo toad (fig. 1).
Most of the areas within FHL that are heavily infested with yellow starthistle occur in or near arroyo toad habitat. In order to apply clopyralid within or adjacent to habitat of the endangered arroyo toad, the federal Endangered Species Act requires formal consultation between FHL and the U.S. Fish and Wildlife Service. Primary concerns that need to be addressed in the consultation are the potential for aerial applications of clopyralid to drift into streams and creeks, and the lack of information on the toxicological effects of clopyralid on amphibians, particularly toads.
Fig. 1. Major yellow starthistle infestations at Fort Hunter Liggett in southern Monterey County. Right, yellow starthistle.
Clopyralid toxicology tests have been conducted on several aquatic species including daphnia, minnows, sunfish and trout (Dow AgroSciences 1998). Clopyralid technical material was found to be practically nontoxic to fish and invertebrate species at LC50 levels (the lethal concentration that kills 50% of test animals) ranging from 104 to 232 parts per million (ppm; milligrams per liter). Formulations containing clopyralid were also nontoxic to fish and invertebrates with LC50 values ranging from 1,100 to 4,700 ppm. Although the toxicity is low for these organisms, there is no clopyralid toxicity data for amphibian species.
To address these concerns, we conducted two separate studies. As a model test animal for arroyo toad, the first study evaluated the susceptibility of larval Fowler's toad (Bufo fowleri) to formulated clopyralid (Transline) in a 96-hour static toxicity test. In a second experiment, we monitored herbicide drift from an aerial application of clopyralid for yellow starthistle control into adjacent buffered vernal pool and stream sites within a military use area on FHL. On the day of application, wind conditions were light (0 to 5 miles per hour [mph]) and blowing from the treated site toward the monitored creek. As such, this study provided an excellent opportunity to evaluate the drift potential of clopyralid into adjacent water sources.
Although the drift study took place within the context of a larger-scale yellow starthistle management study, the objectives of these studies were to (1) determine the effectiveness of a 98-foot (30-meter) buffer between the treatment perimeter and the adjacent stream; (2) monitor clopyralid aerial application for drift into adjacent water bodies; and (3) determine the toxicological effect of the formulated clopyralid solution on larval Fowler's toad.
Treatment location and parameters
The treatment site was located in Training Area 15, a 4,940-acre multiuse site at FHL. Within this area, an 1,110-acre grassland infestation of yellow starthistle was aerially treated by helicopter on the morning of March 19, 2001, with 6 ounces clopyralid (Transline) per acre (2.25 ounces a.e. clopyralid per acre plus 0.125% non-ionic surfactant [First Choice]) at a spray volume of 7 gallons per acre. The helicopter was equipped with a 32-foot single boom approximately 75% of the rotor length and CP 0.078 30-degree deflector nozzles at 9-inch spacings. The application parameters produce a coarse spray per ASAE S-572 and USDA-ARS nozzle testing. Air speed was 70 mph and application altitude was 6 to 7 feet with a maximum of 10 feet in areas with no trees and 40 to 50 feet in areas with oak trees.
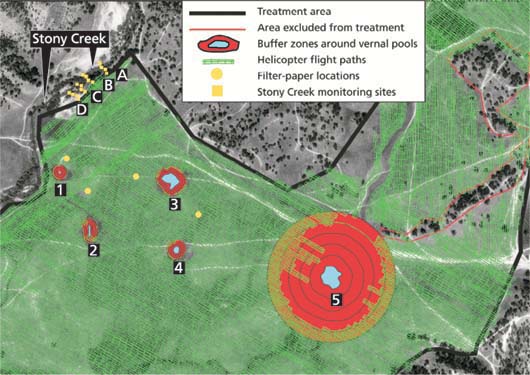
The treatment was applied under a wind speed of 0 to 5 mph. The treatment site was adjacent to Stony Creek, an intermittent stream flowing through the center of Training Area 15 at the time of application. The stream is located south of Lower Stony Reservoir and is a tributary to the Nacimiento River. The creek flows along the northwest boundary of the treatment area. The flowing water and land-based drift-monitoring sites were located along a 1,148-foot (350-meter) section of Stony Creek (fig. 2).
ArcView geographic information system (GIS) software was used to map treatment sites into shape files (NAD 1983 projection) for use with an on-aircraft Trimble navigational global positioning system (GPS) to help the pilot identify buffer zones and the edge of the treatment area. All application passes were recorded using GPS.
The arroyo toad is on the federal endangered species list, and is found at Fort Hunter Liggett. In the study, larval toads had a high tolerance to cloypyralid, and there was little risk of drift into aquatic areas when the herbicide was applied properly from the air.
Within Training Area 15 there are five standing-water pools (fig. 2) exhibiting two or more definitive vernal pool characteristics: hydrology, geology, period of inundation or associated obligate wetland plant species. During spring the pools ranged in size from 0.012 acres (523 square feet/50 square meters for pool 1) to 1.6 acres (69,696 square feet/6,500 square meters for pool 5) and 8 to 15 inches deep, with an average depth of 10 inches. Pool 5, the largest, supports larvae of hybrid California-eastern tiger salamander (Ambystoma sp.)(E.R. Clark, U.S. Fish and Wildlife Service, personal communication). A 98-foot (30-meter) buffer was established along Stony Creek and around vernal pools 1 to 4. This represented the typical distance between the water source and the beginning of the yellow starthistle infestation and was considered a sufficient safety buffer to minimize contamination of the water system. In one exception, however, a 656-foot (200-meter) buffer was established around vernal pool 5, due to tiger salamander larvae.
Before the herbicide treatment, but on the same day, control water and filter-paper samples were taken within the treated areas, in the vernal pools and within Stony Creek. On the day of application, winds were light (< 5 mph) from the southeast direction of the treated area toward the wetland sites. Samples were taken again following the herbicide application.
Monitoring parameters
Samples were placed in certified sterile 1,000-milliliter (ml) jars with Whatman 24-centimeter-diameter (circular) filter paper (MVTL Laboratories, New Ulm, Minn.). Water samples from Stony Creek were taken at eight time periods (three replicates per timing). A total of twenty-four 1,000 ml water samples were collected before treatment and then at 1, 2, 3, 4, 5, 10 and 15 minutes after the perimeter application pass closest to the stream. Vernal pool water samples were taken 1 hour after application. A 98-foot (30-meter) buffer was established between Stony Creek and the spray zone along approximately 3,937 feet (1,200 meters) of the east bank of Stony Creek as it runs adjacent to the treatment area (fig. 2). Four transects approximately 246 feet (75 meters) apart were established perpendicular to the creek. Transects ran from the water edge to the buffer/spray zone interface (fig. 2). Filter disks (Whatman 24-centimeter-diameter [circular] filter paper) were staked at 33-foot (10-meter) intervals along the four transects. The filter papers were pinned to the ground with three or four 10-inch bamboo skewers pushed through the paper. All filter-paper samples were folded and inserted into 500 ml sterile sample jars. Samples were retrieved 30 to 60 minutes after the area was treated.
Filter disks were also placed at four random locations within the treatment zone before the herbicide application to determine clopyralid concentrations. Eight samples were placed in the field. Four samples were collected just prior to aerial application to serve as untreated control measurements and an additional four filter disks were retrieved 30 to 60 minutes after treatment. The disks were folded and inserted into sample jars that were quickly sealed and packed in dry ice; water and filter-paper analyses were conducted by MTVL Laboratories in New Ulm, Minn. The laboratory's method (specifics available upon request) can detect clopyralid residues in water down to the lowest validated level of 0.05 parts per billion (ppb; 0.05 μg/L); the lower detection limit for the filter-paper samples was 0.00125 μg.
Fig. 2. Monitoring sites and aerial clopyralid applications at Fort Hunter Liggett. In upper left, Stony Creek transects A to D were monitored from the edge of the creek to 98 feet away (30 meters; edge of treatment area). Control samples on filter paper were taken from within the treatment area. Vernal pools 1 to 4 are shown with 98-foot (30-meter) buffers; vernal pool 5, the California tiger salamander breeding pool, has a 656-foot (200-meter) buffer. Helicopter flight paths are shown in relation to vernal pools and buffers zones.
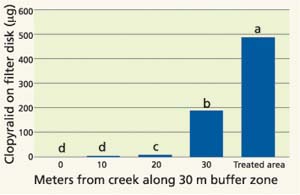
Fig. 3. Clopyralid detected within the treatment zone and 10-meter intervals along a 30-meter buffer between Stony Creek and on treatment area. Values reported at μg/filter disk; detection limit was 0.00125 μg. Bars with different letters represent significant differences at 95% probability.
Clopyralid drift to Stony Creek
Based on an application rate of 6 ounces clopyralid per acre, the expected concentration of clopyralid on each 9.5-inch- (24-centimeter-) diameter disk was 710 μg. However, in the four filter-paper disks within the treatment area, the average concentration of recovered clopyralid was 487 μg (standard deviation = 44 μg)(fig. 3). This represents 69% recovery from the expected clopyralid concentration, which could be due to calibration error, application variability or incomplete extraction in the laboratory evaluation.
The application of clopyralid was mapped using GPS (fig. 2). The GPS spray pattern corresponded closely with the designated buffer zone in the monitored site. The recovery of clopyralid dropped rapidly at greater distances from the treatment zone (fig. 3). At the edge of the treatment area (98 feet [30 meters] from the stream bank) the detected level of clopyralid was 61% lower than within the treatment zone. At 66 feet (20 meters) from the stream, clopyralid was reduced by 98.7% and at the stream bank (approximately 98 feet [30 meters] from the treatment area) only 0.1% of the clopyralid concentration of treatment rate was detected. Clopyralid was undetectable in all samples taken in Stony Creek at all monitoring times (pretreatment and 1, 2, 3, 4, 5, 10 and 15 minutes after application). This suggests that a 98 foot (30-meter) buffer is adequate to prevent clopyralid drift into adjacent water systems, even in the presence of a 5-mph wind blowing from the treatment zone toward Stony Creek.
The drift potential of aerially applied clopyralid was tested in and around vernal pools and streams at Fort Hunter Liggett, where yellow starthistle infests more than 20,000 acres.
Clopyralid drift to vernal pools
Clopyralid was not detectable in vernal pools 1 and 4. The spray pattern from the GPS monitoring indicated that the buffer in vernal pool 1 was not treated (fig. 2). The average depth of the vernal pools was estimated to be approximately 10 inches (25 centimeters). Based on this estimate and the recovery value of clopyralid in the treatment zone (487 μg per disk), it was determined that a direct application of the herbicide into a vernal pool would yield an average clopyralid concentration of 42.0 ppb. In vernal pool 4, a portion of the northwest end of the buffer was accidentally treated, but this was in the downwind side and, consequently, no clopyralid was detected in the pool. In vernal pools 3 and 5, clopyralid was found, but only at the lowest detection limit (0.05 ppb), representing 0.1% of that expected from a direct application into the water body.
The buffer of vernal pool 3 was not treated, but the buffer zone of vernal pool 5 was accidentally encroached on a number of passes and applications were made as close as 131 feet (40 meters) from the vernal pool. Because of the potential presence of the tiger salamander, this vernal pool was supposed to have a 656-foot (200-meter) buffer.
Despite the encroachment, very little clopyralid was detected in the pool. Applicator error in vernal pool 2 accounted for herbicide treatment nearly at the waters edge. In this area, the pilot encroached into the buffer area upwind of the vernal pool. As a result, the concentration of clopyralid in this vernal pool was the highest of all the pools monitored (0.25 ppb). This represented a concentration of clopyralid that was only 0.6% of what would be expected with a direct application in the pool. Overall, the amount of clopyralid found in vernal pools was only 0.2% of that expected with a direct application over water.
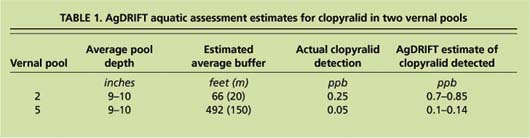
A simulation drift model was used to compare field results with expected results of herbicide drift. Vernal pools 2 and 5 were chosen for the a EPA/SDTF (Environmental Protection Agency/Spray Drift Task Force) AgDRIFT model (Bird et al. 2002) because there was some deposition related to buffer encroachment. Input to the model used the clopyralid rate (6 ounces per acre), application parameters (2.5 to 4 mph wind speed and 8-foot spray height) and the pool's depth and size. The model does not consider wind variation and drift mitigation due to canopy foliage during application. Buffers were visually estimated from data provided through GPS maps and are therefore estimated averages. The model over-predicts deposition from actual field results (table 1) and is expected to be conservative for EPA assessments, but the differences between the model and field results are considered within reasonable environmental variations.
Toad toxicology experiment
Because the arroyo toad is a federally listed endangered species it could not be used in toxicology tests; instead, we used a related species (Fowler's toad) as a model. The California Department of Fish and Game's Aquatic Toxicology Laboratory in Elk Grove conducted toxicity testing. Larval toads were exposed to formulated clopyralid (Transline) in 96-hour static toxicity tests following established laboratory procedures (DFG 2000, 2001) and federal testing guidelines (US EPA 1993).
Larval toads (17 days old) were purchased from Northeastern Aquatics (Rhinebeck, N.Y.) and were delivered to the laboratory in good condition. After acclimation at 36°F (2°C) for 96 hours, 10 larval toads were loaded into 1-liter Pyrex test cups containing 250 ml of the test solution. Excluding the control, there were six test solutions ranging from 151 to 1,030 ppm (mg/L)(measured as technical clopyralid)(table 2). After the tadpole loading, the test cups were placed in environmental chambers. Random numbers were used to select the location of test cups within the chambers. Chamber temperatures were maintained at 72°F ± 2°F (22°C ± 1°C) for the duration of the test with a photoperiod regime of 16 hours light and 8 hours dark.
To ensure a constant concentration of clopyralid, the treatment solutions were replaced on the second test day. Water quality (conductivity, temperature, pH and dissolved oxygen) was monitored daily in the test cups. Alkalinity, total hardness and toxicant concentrations were determined at the initiation of the experiment and 2 days after exposure in the original and replaced solutions. Test chambers were checked daily for tadpole mortality. Dead larvae were removed from the test chambers upon discovery.
Tadpole survival in the untreated control was 100% (table 2). Similarly, no larvae mortality was observed at the lowest treatment concentration of 151 ppm clopyralid. Fowler's toad larvae did not survive in test-solution concentrations of 538 ppm or higher. The 96-hour LC50 value for larval Fowler's toads was 413 ppm clopyralid. The U.S. EPA designates LC50 values greater than 100 ppm as “practically nontoxic” to nontarget aquatic organisms (Zucker 1985).
Using the highest concentration where no toxicological effect was observed (151 ppm) and assuming that under a worst-case scenario clopyralid was accidentally applied directly over water (10 inches [25 centimeters] deep) at the highest registered rate (10.7 ounces per acre) the clopyralid concentration in water would be 0.11 ppm. This would provide a safety factor greater than 1,370 for larval toads. When the clopyralid concentration obtained in the drift experiment is used (0.00025 ppm), the safety margin for larval toads is increased to greater than 600,000 (151 ppm/0.00025 ppm).
Kyle Emery tests tadpoles in an environmental chamber, which maintains constant temperature and humidity as well as a light-dark regime.
These findings provide direct evidence showing minimal drift movement of clopyralid off-site, despite an aerial application and a slight wind in the direction of the sensitive site. In addition, this is the first report demonstrating a high tolerance to clopyralid in larval toads, suggesting that a 98-foot (30-meter) buffer between the treatment zone and water sources is sufficient to provide a reasonable margin of safety to aquatic organisms, presumably including toad species.