All Issues
Beneficial insects move from flowering plants to nearby crops
Publication Information
California Agriculture 52(5):23-26. https://doi.org/10.3733/ca.v052n05p23
Published September 01, 1998
PDF | Citation | Permissions
Abstract
Marking studies demonstrated that lady beetles, lacewings, syrphid flies and parasitic wasps fed on nectar or pollen provided by borders of flowering plants around farms; many insects moved 250 feet into adjacent field crops. Studies using the elemental marker rubidium also showed that syrphid flies, parasitic wasps and lacewings fed on flowering cover crops in orchards and that some moved 6 feet high in the tree canopy and 100 feet away from the treated area. The use of nectar or pollen by beneficial insects helps them survive and reproduce. Therefore, planting flowering plants and perennial grasses around farms may lead to better biological control of pests in nearby crops.
Full text
Entomologist Andrew Corbett sprays a rubidium solution onto toothpick weed (A. visnaga) at Lindell Farms in Fresno County to track the movement of beneficial insects.
California farmers are planting strips or borders of flowering plants and perennial grasses, termed insectary plantings, around their farms to attract beneficial insects and thus get better biological control of pests in their crops. The idea behind these plantings is that many adult beneficial insects feed on nectar or pollen as a sole food source or to supplement their diet during periods of prey scarcity. These critical nutrients help them survive and increase their egg-laying ability. To the extent that these food resources lead to more beneficial insects around farms, insectary plantings may result in greater biological pest control.
While these plantings seem like a good idea, there are few data to support their application as an effective biocontrol tool. The objective of this study was to determine if beneficial insects feed on insectary plants and then move into associated crops.
Insectary plants
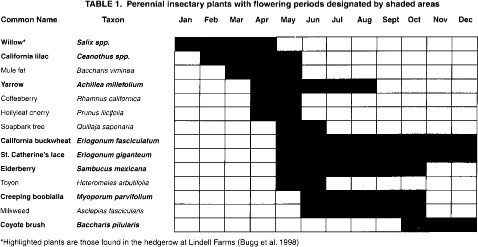
Insectary plants are those that produce nectar or pollen and attract beneficial insects. These include perennial and annual plants (tables 1 and 2) and perennial grasses such as deergrass (Muhlenbergia rigens), purple needle-grass (Nassella pulchra), blue wild rye (Elymns glaucus), meadow barley (Hordeum brachyantherum), California brome (Bromus carinatus) and Yolo slender wheatgrass (Elymus trachycaulus majus). Insectary plantings that contain a variety of plants with different flowering periods provide a year-round food supply for beneficial insects.
Using rubidium to track insects
Rubidium (Rb) was used to document the feeding by beneficial insects at insectary plantings and their movement into adjacent crops. This element substitutes for the potassium in plant and insect tissues. Tests have shown that at low concentrations rubidium does not have any detectable effects on longevity or behavior of most insects (Stimmann et al. 1973). It can also be easily tracked in biological systems through the use of a flame emission spectrophotometer.
Beneficial insects were labeled by injecting or spraying solutions of rubidium chloride (RbC1) into or on insectary plants. The insects obtained the label through feeding on Rb-rich nectar and pollen sources that concentrated in the plant. Because Rb occurs naturally in plant and insect tissues at fairly low and uniform levels, relatively small increases in Rb result in a measurable label at levels above the natural background concentration normally found in insects (Berry et al. 1972).
At the almond orchard site, lacewings, syrphid flies and parasitic wasps moved up to 100 feet away from the insectary plantings.
Three field sites
From 1994 to 1995, we conducted studies at three field sites where insectary plantings are being used by growers. The first site was at Hedgerow Farms in Yolo County, with fourteen 10-year-old California lilac (Ceanothus ‘Ray Hartman’) bushes located directly across from a wheat field. The second site was at Lindell Farms in Fresno County, with a 2-year-old, 20-foot-wide-by-660-foot-long hedgerow of perennials (table 1) and annual toothpick weed (Ammi visnaga). This insectary planting was surrounded by organic vegetables including beans, corn and tomatoes, and a small orchard of peaches and walnuts. The third site was in an almond orchard in Merced County, where a grower planted a mix of winter insectary annuals in 1 out of every 10 tree rows (table 2).
Rubidium applied to plants
We injected RbC1 into the California lilac plants at Hedgerow Farms. This was done once before bloom, by drilling one or two holes into each main branch at the base of the plant and injecting 7 milliliters of RbC1 solution (at a very high concentration of 0.25 gm per mL) into each hole with a syringe. Injections increased the Rb content of plants by about 10 times the naturally occurring background levels. The Lindell Farms site was sprayed with RbC1 at 3,000 ppm about every 2 weeks from June until August. At the almond orchard site, four cover crop strips (6 ft by 1,000 ft) were sprayed with RbC1 at 3,000 ppm about every 2 weeks from May to July. RbC1 sprays increase the Rb content by 200 times the naturally occurring background levels.
Sampling beneficial insects
Following RbC1 applications, we collected beneficial insects from the insectary plants and the associated crop using 6-inch-by-12-inch sticky cards of similar color to standard yellow white-fly traps. Only those beneficial insects believed important to pest management in the crops were collected for Rb analysis (table 3).
At Hedgerow Farms, we suspended 8 traps in the canopy of individual California lilac shrubs. In the adjacent wheat, we stapled 20 traps onto wooden stakes at the level of the crop at distances of 50 feet, 150 feet, 250 feet and 450 feet from the California lilac. We changed all of the traps every 2 weeks from April until May.
At Lindell Farms, we placed 10 traps on wooden stakes every 50 feet, adjacent to and just below the height of the insectary plants. In the adjacent vegetable crops, 20 traps were stapled onto wooden stakes below the level of the crop at distances of 20 feet and 250 feet from the insectary plants. In the adjacent orchard, 20 traps were placed 6 feet high in individual trees at 50-foot intervals, from 20 feet to 250 feet away from the hedgerow. Traps were changed every 2 weeks from June to August. We collected Hyposoter wasps using a handheld aspirator, as we did not find them on sticky traps.
At the almond orchard site, we placed 36 traps 6 feet high in individual trees (24 ft by 24 ft spacing) at 1, 3 and 5 tree rows from the insectary mix strips. Traps were changed every 2 weeks from May until July.
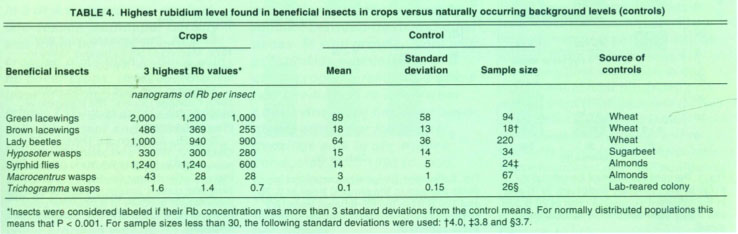
We removed the beneficial insects from the traps, then identified and analyzed them for Rb content. Similar beneficial insect species were collected from „control“ fields, located more than 2 miles away from treated hedgerows, to determine the naturally occurring background Rb content. Trichogramma controls were obtained from a laboratory colony. These data provided the criteria for deciding if an individual beneficial insect captured within the insectary plantings and associated crops was labeled (table 4).
Analysis of samples
Insects were chemically digested and analyzed for Rb content using standard methodologies (Berry et al. 1972). Insects were considered labeled if their Rb concentration was more than three standard deviations greater than the control means. In this way, the probability of incorrectly identifying an insect as labeled with Rb is very low (less than 1%).
Labeling of beneficial insects
Hedgerow Farms. In the California lilac, 67% (sample size of n = 15) of the green lacewings, and 40% (n = 10) of the lady beetles were labeled with Rb, following injections with RbC1. In the adjacent wheat, 2% of the green lacewings were labeled at 50 feet and 2% were labeled at 450 feet (n = 47). We did not find any labeled lady beetles in the adjacent wheat.
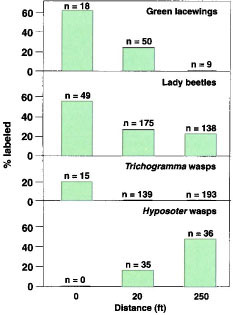
Lindell Farms. RbC1 sprays in the insectary hedgerow also resulted in marking of beneficial insects (fig. 1. In the insectary plantings, 61% of the green lacewings were labeled, plus 20% of the Trichogramma, and 55% of the lady beetles. We did not find any labeled Hyposoter wasps in the insectary plantings, even though these wasps are known to feed on nectar of flowering plants. We suspect that these wasps were visiting the plants in a manner that escaped our detection, possibly due to the time of day or their foraging behavior.
In the adjacent vegetables (fig. 1), 24% of the green lacewings were labeled 20 feet from the hedgerow, but none were labeled at 250 feet. For lady beetles, 27% were labeled at 20 feet and 23% were labeled at 250 feet from the hedgerow. For Hyposoter wasps, 17% were labeled at 20 feet and 47% were labeled at 250 feet from the hedgerow. None of the Trichogramma were labeled in the nearby crops.
In the adjacent orchard, 10% of the green lacewings were labeled at 20 feet (n = 31) and 33% (n = 3) were labeled at 150 feet from the hedgerow. No labeled green lacewings were found in the trees at any other distances (n ≪ 3 for all these samples). Ten percent of the lady beetles (n = 10) and 2.5% (n = 40) of the Trichogramma were labeled at 20 feet from the hedgerow.
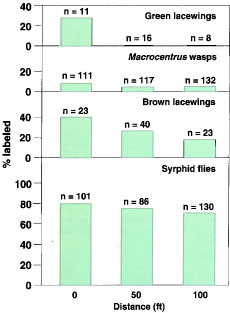
Almond orchard site. RbC1 marking of the annual insectary mix also showed labeling of beneficial insects (fig. 2). In the trees, 27% of the green lacewings were labeled adjacent to the insectary mix, but none were labeled further away. For brown lacewings, 39% were labeled next to the insectary mix, 28% were labeled at 50 feet and 17% were labeled at 100 feet. For syrphid flies, 81% were labeled adjacent to the insectary mix, 74% were labeled at 50 feet, and 70% were labeled at 100 feet. For Macrocentrus wasps, 8.1% were labeled next to the insectary mix, 4.3% were labeled at 50 feet, and 4.5% were labeled at 100 feet.
Underestimation of numbers
Although the percentage of labeled beneficial insects was low in some samples, these data probably underestimate the numbers of beneficial insects that are using the insectary plants. Our laboratory studies showed that green lacewings need to feed on Rb-marked floral resources for up to 24 hours to acquire detectable levels of Rb. If beneficial insects are feeding on both marked and unmarked plants in the field, the impact of the Rb label will likely be diluted. For example, at Hedgerow Farms, where we had low levels of labeling, there is an abundance of floral resources other than our Rb-marked California lilac for beneficial insects to feed on. In addition, our laboratory studies showed that green lacewings excrete the label within 24 hours.
TABLE 4. Highest rubidium level found in beneficial insects in crops versus naturally occurring background levels (controls)
Scientists found that 70% of the syrphid flies in almond trees were marked with rubidium 100 feet away from the insectary planting.
Fig. 1. Percent Rb-labeled beneficial insects in the insectary hedgerow (0 ft) and in the adjacent vegetable crop at 20 ft and 250 ft (n = total number of insects captured).
Fig. 2. Percent Rb-labeled beneficial insects trapped in the almond trees at 0, 50 and 100 ft from the Rb-labeled insectary cover crop mix (n = total number of insects captured).
The results of this study demonstrate that beneficial insects feed on nectar or pollen provided by insectary plants, and that they move into associated crops. Because many Rb-labeled individuals were trapped 250 feet from the insectary plants, our data suggest strips or borders of flowering plants may help beneficial insects — even at long distances. The survival of many beneficial insect species depends on nectar or pollen so adding insectary plantings to the agricultural landscape may enlarge their populations and increase their activity in crops.