All Issues
Fungal pathogen controls thrips in greenhouse flowers
Publication Information
California Agriculture 52(3):32-36. https://doi.org/10.3733/ca.v052n03p32
Published May 01, 1998
PDF | Citation | Permissions
Abstract
Western flower thrips cause considerable losses in a wide range of agricultural crops by feeding on leaves and fruit, laying eggs in fruit and transmitting diseases. Repeated pesticide application is currently the only method that reduces populations to acceptable levels. Biological control efforts have focused on using predators and have been largely unsuccessful. However, entomopathogenic fungi could also be used as biological controls for western flower thrips, Laboratory and field trials show that commercial formulations of Beauveria bassiana (GHA strain) can infect and reduce western flower thrips numbers in greenhouse floriculture crops, thus demonstrating its potential as an alternative to conventional pesticides.
Full text
The western flower thrips is one of the most significant pests of cut flower production in California. Thrips feed on flower petals, scarring them and causing aesthetic damage that can render the flowers unmarketable. For many flower crops, most of the pesticide sprays applied are to target thrips. The industry estimates pest control costs (labor and materials) to be 7.5% of total product costs. It is difficult to quantify the loss of flower production to thrips because the state doesn't keep statistics on it.
Infestations also cause considerable losses in a wide range of agricultural crops such as tomatoes, peppers, and stone fruits, by directly feeding on foliage and fruit and through ovipositional injury to fruit. In addition, western flower thrips serves as a vector for both tomato spotted wilt virus and impatiens necrotic spot virus, which attacks a wide range of floral and vegetable crops.
Current management tactics for western flower thrips (WFT), Frankliniella occidentalis, in floricultural production rely predominantly on repeated pesticide applications. In many situations, growers apply pesticides at 5-to-10-day intervals to reduce populations to acceptable levels. Such intensive use of pesticides has resulted in the widespread development of pesticide-resistant WFT in greenhouses. Efforts to reduce reliance on chemical controls have focused on biological control using predatory mites in the genera Amblyseius and Hypoaspis and predatory bugs in the genus Orius.
However, biological control alone has not succeeded in reducing thrips populations in floriculture crops to acceptable levels. The high demand for aesthetic quality and the problem associated with virus transmission make achieving successful control with natural enemies very difficult. Because the price of the commodity is linked to the aesthetic quality, growers tend to have a low tolerance for damage.
Another potential alternative to traditional pesticides is the use of entomopathogenic fungi, which grow on thrips and other arthropods. Naturally occurring fungal pathogens are lethal to many arthropod species. When the temperature and relative humidity are optimal, a large proportion of an insect population can become infected with the fungus, resulting in significant reductions in insect population size; this is called an epizootic.
The common occurrence of natural fungal epizootics has led to attempts to exploit fungi as a method of pest control in field and greenhouse crops around the world. To date, commercial use has been limited by technical difficulties with the mass production and shelf life of conidia (spores), formulation and variable performance among fungal species and strains, as well as the demanding environmental conditions required for fungal infection. Recently, however, advances in fermentation and formulation technologies and better isolation of infectious species and strains have led several manufacturers to begin commercial production of fungi for pest control.
Western flower thrips adult female showing the sporulating fungi of Beauveria bassiana, indicating the thrips was killed by the fungal infection.
Fungal pathogens have several characteristics that make them ideal candidates for alternatives to chemical pesticides. Often fungi are relatively host specific, have low mammalian toxicity, can be cheaply mass-produced on artificial media and can infect a high proportion of the target population. In addition, fungi can be readily formulated and applied using standard spray equipment.
High temperature and relative humidity are required for most fungi. However, some newly discovered species and strains appear capable of infections over a wider range of environmental conditions commonly found in the greenhouse (60° to 85°F, 50 to 100% relative humidity). Although different life stages of fungi can be applied, the conidiospores (spores) are most often used as the agent of control. Insects can get spores on them either from direct spraying or from contacting plant foliage that has been sprayed. After becoming, attached to a susceptible host, a spore grows a germ tube that penetrates the insect's cuticle. This enables the fungus to feed on the host's body, ultimately killing the insect.
Warm temperatures and relatively high humidities make greenhouses ideal environments for using fungal pathogens. Here, we present the results of laboratory and greenhouse trials designed to evaluate the potential of using a commercially produced strain of Beauveria bassiana for control of western flower thrips in greenhouse floriculture crops. Two commercial products containing the fungus are currently available: BotaniGard (a wet-table powder [WP] or emulsifiable oil [ES] that is produced by the Mycotech Corp. of Butte, Montana) and Naturalis-O, (produced by Troy. Biosciences Inc. in Lake Placid, Florida). In the experiments reported here, we used the BotaniGard WP and ES formulations of the fungus.
Laboratory and field trials
Laboratory trials.
We conducted laboratory trials to assess the effectiveness of B. bassiana against mixed ages of adult male and female WFT on rose foliage at different spore concentrations under controlled temperature and relative-humidity conditions. Concentrations of 0.1, 0.45, 0.9 and 1.8 grams of spores (4.4 × 1,010 spores/gram) per 100 milliliters water plus a spreading agent (0.3% v/v, Silwet L-77) were tested on rose foliage against WFT and compared to WFT treated with the spreading agent alone. In addition, tests were conducted to compare the influence of three relative humidities (60%, 75% and 90%) on WFT mortality at two spore concentrations (0.9 and 1.8 grams spores per 100 mL water). WFT were confined on rose foliage within small cardboard cartons with a clear petri dish lid. Each carton represented an experimental replicate. Four replicates containing 20 to 50 adult WFT at each concentration and relative humidity were compared.
Approximately 0.6 milliliters of B. bassiana spore suspension was applied to rose foliage and WFT within cartons using a laboratory spray tower. Cartons were held in environmental chambers at 78.8°F (26°C) at one of the three relative humidities. The temperature and relative humidity in the vented cartons closely approximated those in the environmental chambers (within 3.6°F [2°C] and 5% RH). Every 24 hours for 7 days, we counted the number of dead WFT in each carton. Differences in the proportion WFT mortality among treatments were analyzed by ANOVA and mean comparisons between treatments were performed using Dunnett's method at P = 0.05. Probit analysis was used for estimating the dose-mortality relationships.
Caged rose trials.
Initial tests evaluating the efficacy of a B. bassiana wet-table powder (WP) against WFT adult females on flowering plants were performed on caged rose buds. In a commercial greenhouse, 16 rose bud replicate cages were used within a randomized complete block design. We sprayed 8 of the rose bud replicates with B. bassiana WP at 1 pound of formulation per 100 gallons of water plus a spreading agent (as discussed above) and sprayed the other 8 with water and spreading agent only, then allowed them to dry. Flower buds were then enclosed with a Mylar tube cage with a mesh screen at either end to allow for ventilation. We aspirated 12 to 15 adult female WFT from carnation flowers and released them into each of the cages.
After 7 days, cages were pruned from the rose bushes and returned to the laboratory and placed in a conventional freezer to kill WFT within the cages. Because of the high rate of activity of WFT, it is necessary to inactivate WFT to obtain accurate counts. We then dissected the flower buds and recorded the number of adult and larval WFT. The WFT recovered from cages were then emersed in alcohol to kill spores on the insect cuticle and plated on a selective agar medium to determine the degree of fungal infection within WFT among treatments.
Commercial greenhouse trials
Field studies were conducted at two locations: Watsonville and Half Moon Bay. Trials compared the performance of the B. bassiana wettable powder (WP) and an emulsified oil (ES) formulation of B. bassiana
Carnations.
The Watsonville trial was conducted against WFT in a 40,000-square-foot greenhouse in three carnation cultivars — ‘Elegance’, ‘Etna’ and ‘Bagatel’. Test plots consisted of 3 treatments with 6 replicates for each treatment (approximately 725 square feet per plot for a total of 13,050 square feet). Six plots were treated with B. bassiana wettable powder (WP) formulation (1 lb/100 gal), 6 plots were treated with B. bassiana emulsifiable oil (ES) formulation (2 qt/100 gal) and 6 plots were untreated controls. The mean number of WFT residing in the carnations was counted just prior to the beginning of the trial. Two applications were made 8 days apart, the first on Nov. 28 and the second on Dec. 6, 1995. To compare mean WFT densities among plots, we sampled 10 to 15 fully opened carnation flowers from each plot on Dec. 6 and 13.
Roses.
The Half Moon Bay trial was conducted against WFT within a 50,000-square-foot, commercial fresh-cut rose greenhouse in two rose cultivars, ‘Royalty’ and ‘Caramia’. The trial consisted of three treatments, the first was B. bassiana WP (1 lb/100 gal), the second B. bassiana ES (2 qt/100 gal) and the third was an emulsifiable oil carrier only as the control (2 qt/100 gal).
The trial was designed as a randomized complete block design using 12 test plots of approximately 1,185 square feet each. Each of the two blocks consisted of 6 plots; treatments were randomly assigned to all plots within each block. There were four replicates per treatment. A one-row buffer was maintained between each treatment plot to minimize drift from spray applications. We monitored WFT densities just before and after treatment by sampling 10 rose flowers per plot. Four treatment applications were made at 7-day intervals beginning on April 3, 1996, and ending on April 24, 1996.
For both the carnation and rose field trials, we bagged flower samples individually to prevent movement of WFT between samples. Samples were returned to the laboratory and kept in a conventional freezer until processing. We dissected WFT from rose or carnation flowers and recorded the total number, For the rose trial, 5% chlorine bleach was used to kill any spores on the insect cuticle and plated the WFT on selective agar to determine the rate of fungal infection. Differences in WFT mortality among treatments in the caged rose and commercial trials were analyzed by ANOVA and mean comparisons between treatments were performed using Tukey's mean separation test at P = 0.05.
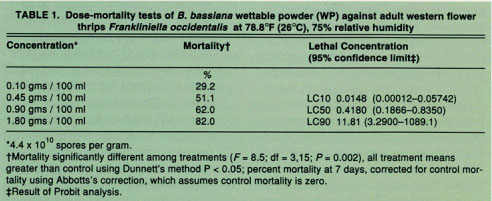
TABLE 1 Dose-mortality tests of B. bassiana wettable powder (WP) against adult western flower thrips Frankliniella occidentalis at 78.8°F (26°C), 75% relative humidity
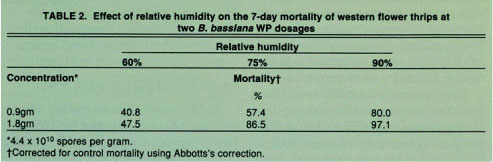
TABLE 2 Effect of relative humidity on the 7-day mortality of western flower thrips at two B. bassiana WP dosages
Reductions in thrips numbers
Laboratory trials.
Results of the laboratory trials revealed that B. bassiana WP applications caused significantly greater WFT mortality at all concentrations when compared to controls at 78.8°F (26°C) and 75% relative humidity (table 1). WFT mortality did not exceed 90% for any single concentration tested. Therefore, we estimated the optimal concentration resulting in 50% WFT mortality. Probit analysis determined that B. bassiana WP killed 50% of the WFT population at a concentration of 0.42 grams per 100 milliliters of water (table 1). This concentration is the equivalent of applying 20 trillion spores per acre (1 lb B. bassiana WP). Doubling the concentration first to 0.9 grams/100 milliliters and then again to 1.8 grams/100 milliliters, did not double the mortality rate, indicating that the initial concentration of 0.45 grams/100 ml yielded close to the maximum mortality at this temperature and relative humidity.
Relative humidity tests revealed that significant mortality occurred at all relative humidities tested; however, the degree of mortality was shown to increase with increasing humidity (table 2). Relative humidities in California greenhouses vary depending on geographical region and greenhouse type but typically range between 50 and 100% within a 24-hour period. The laboratory results confirmed that humidity is an important factor influencing the degree of WFT control but use of either formulation should significantly reduce WFT numbers under California greenhouse conditions.
Caged rose trial.
Results of the caged trials confirmed that B. bassiana applications to rose buds infested with WFT can result in significant reductions in WFT numbers. The magnitude of the reduction was similar to that found in the laboratory bioassays.
Initial WFT counts within cages revealed that control cages averaged 16.5 +/- 3.35 WFT and that cages treated with B. bassiana WP averaged 11.5 +/- 2.58 (fig. 1). Differences in the average number of WFT within cages were not significantly different between treatments (P > 0.05). However, at the time of collection it was observed that most WFT in the treated cages were dead while most in the control cages were living. By plating all WFT cadavers on agar medium, we determined that B. bassiana infection was present in 42.4% of the WFT in the B. bassiana-treated cages whereas only 8.3% in control cages were infected.
Fig. 1 Mean number of fungus-infected and healthy western flower thrips in caged rose buds in untreated control and B. bassiana WP-treated cages. Treatment means separated by different letters are significantly different (Tukey's mean separation test, P < 0.05).
After subtracting the number of fungus-infected individuals from the total number of thrips, the net number of WFT that were uninfected was 6.6 +/- 1.67 for the B. bassiana-treated cages and 15.1 +/- 3.25 for the control cages. Therefore the number of surviving WFT in the cages were 56% lower in the B. bassiana-treated cages relative to the control cages. Differences in the remaining uninfected WFT between treatments were significant (F = 5.98; df = 2, 15; P = 0.029).
Commercial carnations.
Prior to treatment applications, Mean WFT in carnation flowers among plots were not significantly different (fig. 2). After the first treatment, WFT numbers increased in all plots presumably due to WFT moving into the greenhouse. For the first postspray sample period, WFT numbers were significantly lower in the B. bassiana ES plots than in the control plots, but not in the B. bassiana plots. By the second postspray period, WFT numbers had begun to drop in all plots. WFT numbers were significantly lower in B. bassiana-treated plots than in the control plots (fig. 2): WFT numbers were 59% lower in ES plots and 72% lower in WP plots. Fungal infection data for WFT were not available for the carnation trial. During the trial, greenhouse temperatures averaged between 50° and 82°F (10.0°C and 27.8°C) and the relative humidity ranged between 45% and 80% during a 24-hour cycle.
Commercial roses.
Pretreatment WFT numbers showed average WFT densities were approximately even among treatment plots prior to applications (fig. 3). During the first postspray sample period, significantly fewer WFT were detected in the B. bassiana ES plots relative to control plots, but the numbers in the B. bassiana WP plots were not significantly lower than the control. By the second postspray sample, WFT numbers were also significantly lower in B. bassiana-treated plots than in control plots: WFT were 83% lower in ES plots and 75% lower in WP plots. Thereafter, WFT numbers in the B. bassiana plots remained below 1 WFT per rose bud. During the trial, greenhouse temperatures averaged between 60°F and 90°F (15.5°C and 32.2°C) and the relative humidity ranged between 45% and 100% during a 24-hour cycle.
Pretreatment examination of WFT showed that there was a low-level natural infection of a Beauveria species similar to B. bassiana (data not shown). However, morphological differences between the fungal species allowed us to distinguish them. The fungal infection rates during the trial revealed the presence of infected WFT in all treatments, including the controls. However, the rate of infection was significantly greater in the B. bassiana treatments (fig. 4) except for the WP plot on April 17. We suspect that the fungal infection in the control plots was the result of infected adult WFT dispersing from the B. bassiana-treated plots.
Commercial fungus kills thrips
Western flower thrips is a key pest of most floriculture crops. Without viable alternatives for WFT control, the only means to prevent damage is through repeated pesticide applications. Residues from repeated pesticide applications targeting WFT often kill natural enemies of other greenhouse pests and therefore may both contribute to secondary pest outbreaks and interfere with adoption of IPM tactics such as biological control. The lack of effective alternatives to traditional pesticides for WFT control has therefore led to over-reliance on pesticides in greenhouses despite increasing costs and other negative secondary effects. The performance of B. bassiana in this study indicates that fungal pathogens could be substituted for some pesticide applications.
Fig. 2 Mean number of healthy WFT in carnation flowers for untreated controls and B. bassiana WP-treated and B. bassiana ES-treated plots. Treatment means separated by different letters are significantly different (Tukey's mean separation test, P < 0.05).
Fig. 3 Mean number of healthy WFT in rose buds for emulsified oil controls and B. bassiana WP-treated and B. bassiana ES-treated plots. Treatment means separated by different letters are significantly different (Tukey's mean separation test, P < 0.05).
Fig. 4 Mean proportion of fungus-infected WFT in rose buds for emulsified oil controls and B. bassiana WP and B. bassiana ES-treated plots. Treatment means separated by different letters are significantly different (Tukey's mean separation test, P < 0.05).
Laboratory and caged rose trials demonstrated that commercial formulations of B. bassiana can infect WFT and reduce populations in the greenhouse. Reductions were variable, ranging from 50 to 97%. Similar results were seen in both the commercial carnation and rose trials. In a greenhouse setting, it is difficult to prevent infected WFT from contaminating control plots; as a result, the percentage of control achieved with B. bassiana in the commercial trials should be considered conservative estimates.
Fungal infection is sensitive to environmental conditions. Increases in temperature or, particularly, relative humidity can influence the rate of fungal infection and the degree of WFT control achieved. Trials with commercial growers demonstrated that good performance could be obtained under commercial growing conditions that include the temperature and relative humidity regimes typical of greenhouses. Differences in performance between the two formulations were evident in both greenhouse trials. The oil formulation of B. bassiana reduced WFT numbers within the first week while the wettable powder required up to 2 weeks. The reason for this difference is currently being investigated. Our results thus far indicate that while both commercial formulations of B. bassiana could be used to help control WFT in greenhouse flowers, the ES formulation works more quickly. Growers tolerate very few WFT in their flowers; based on these studies, B. bassiana is capable of reducing WFT in flowers to very low levels, generally within growers' tolerable range.
Although the results so far are encouraging, several limitations are of concern. First, fungal pathogens kill host insects more slowly than insecticides and therefore must be applied earlier in the appearance of WFT to maintain the thrips at low levels. Second, under severe immigration pressure, the movement rate of WFT into the crop can exceed the rate of fungal infection and acceptable reductions may not be achieved soon enough to prevent damage. Finally, spores kill insects through direct contact with their hosts, therefore, good coverage is essential to achieve adequate control.
Commercial field trials in rose, poinsettias and chrysanthemums for WFT and whitefly control are currently under way to further develop user guidelines for B. bassiana. These trials are aimed at estimating the optimal field dosage rate, spray intervals and evaluating the combined use of B. bassiana with other insecticides as well as with natural enemies.