All Issues
Pest management alternatives needed: Delaney Clause ruling may trigger pesticide cancellations
Publication Information
California Agriculture 48(1):30-35.
Published January 01, 1994
PDF | Citation | Permissions
Abstract
As a result of a court ruling that the Delaney Clause must be strictly enforced, tolerances for some pesticides on processed foods and raw agricultural commodities may be revoked. According to the clause, no residue tolerances for pesticides shown to induce cancer may remain in processed food. Unless there is a significant change in law or policy, as many as 35 pesticide/commodity registrations will be canceled because EPA's current policy prohibits establishing a raw commodity tolerance if a tolerance on processed food is prohibited. If alternatives are not available, production of specific commodities may suffer.
Full text
Under the Federal Insecticide, Fungicide and Rodenticide Act (FIFRA), all pesticides used in the United States must be registered by the U.S. Environmental Protection Agency (EPA). Pesticide registration is a legal process that allows a chemical to be used for pest management on crops, livestock and land in the United States. Most pesticides leave minute amounts of residues on the crop or other treated site after application. The Federal Food, Drug and Cosmetic Act (FFDCA) requires every pesticide used on food or feed to have a legally established residue tolerance for each crop to which it will be applied or to animal products such as meat and milk where residues may remain. Tolerances are the maximum legal amount of pesticide residues that may remain on agricultural products when they are ready for market. The tolerances are set at levels that provide safety margins to protect public health. (For additional comment on the current tolerance system, see articles beginning on p. 13 and p. 21 .)
Tolerances are based on the percentage of pesticide or its breakdown products that can remain on the product. They are expressed as parts per million (ppm); For example, 1% equals 10,000 ppm; 10 ppm equals 0.001%. Typically tolerances are in the range of 1–10 ppm.
Under FFDCA, tolerances are established for raw or unprocessed agricultural products as they come from the farm. Separate tolerances are set for processed foods, for example those that have been dried or milled. Pesticide tolerances on processed foods are required only when the processing will leave residues that exceed the level allowed on the raw product.
Residues legal 99% of time
Agricultural producers have been very successful in staying within the raw agricultural produce tolerance limits. About 99% of agricultural products delivered to market have residues that are either below the detection limits of current analytical technology or within legally acceptable limits. Based on sampling by the State of California, approximately 1% of agricultural products that reach the market exceed the established tolerance or have residues of pesticides not currently registered for the specific crop. (Additional discussion of sampling programs appears on p. 18 and p. 20. )
Above and right, grapes, potatoes and tomatoes are among the crops whose production might suffer under strict enforcement of the Delaney Clause.
Nationwide produce sampling has consistently shown that when food from the marketplace is analyzed, average residue levels are minuscule. Overall hazards to human health from residues are low. Furthermore, EPA has typically applied a standard of “negligible risk” in regulating potential carcinogens: the regulatory limit for such substances is the amount of pesticide residue that, consumed daily over a 70-year life span, has the potential to cause one additional cancer in a million people.
In establishing tolerances on raw agricultural products, the EPA is required to evaluate and consider both the risks and benefits that will result if the pesticide is registered. Human, animal and environmental risks may be present whenever a pesticide is used. Conversely, there are benefits to both growers and consumers. For growers, benefits typically include more reliable and less costly agricultural production. For consumers, benefits include less costly food, reliable supply, consistent quality and freedom from bacterial, fungal and insect contamination.
Paradox: the Delaney Clause
Under the Federal Food, Drug and Cosmetic Act, pesticide residue tolerances must be set for processed foods. According to Section 409 of this law ( see sidebar, p. 34 , and table 1), pesticides remaining on processed foods are considered to be food additives. Section 409 includes the Delaney Clause, adopted in 1958, which prohibits food additives that have been shown to cause cancer in humans or animals.
FIFRA requires the administrator of the EPA to establish guidelines for tests to determine the cancer-causing potential of pesticides registered in the United States. The tests typically involve feeding large doses of the chemical to two animal species, usually rats and mice, for 2 years. It is difficult to make meaningful extrapolations from high test doses to the minute amounts actually consumed by humans. Such extrapolations may overestimate the potential adverse effects on human health. Regardless, if any statistically significant increase in cancers is found in the test animals, FFDCA prohibits the residues in processed food. Paradoxically the laws do not prohibit the same residues in the raw agricultural products that reach the market.
Because of the paradoxical regulatory situation that exists between pesticide tolerances on raw agricultural commodities and processed foods, the EPA has a policy for coordinated tolerance setting. EPA's coordination policy prohibits establishing a raw agricultural commodity tolerance (Section 408) if a tolerance on processed food is prohibited under Section 409.
EPA's de minimis principle
In the past the EPA has regulated pesticides in processed food on the basis of the de minimis principle of law. This principle has been applied when risk has been considered trivial. Under this principle an administrative agency may avoid applying the terms of a law literally when doing so would lead to scientifically unsound results. When predicted risks from residues are so low that they have been considered negligible, EPA has set tolerances on both raw and processed food for a few pesticides that induced cancer in laboratory animal tests.
The EPA's use of the de minimis principle was challenged in the courts. Recently a federal court held that the Delaney Clause prohibits applying the de minimis principle when setting pesticide tolerances for processed commodities. According to EPA's current policies, once a pesticide tolerance is revoked, the corresponding registration for that use is canceled to prevent illegal residues. As a result of the court's Delaney decision, tolerances for some pesticides on both processed foods and raw agricultural commodities may be revoked. Unless there is a significant change in law or policy, some pesticide registrations will be canceled. The EPA has published a list of 35 pesticide/commodity combinations that might be canceled in the future. This list includes many pesticides used on crops in California (table 1).
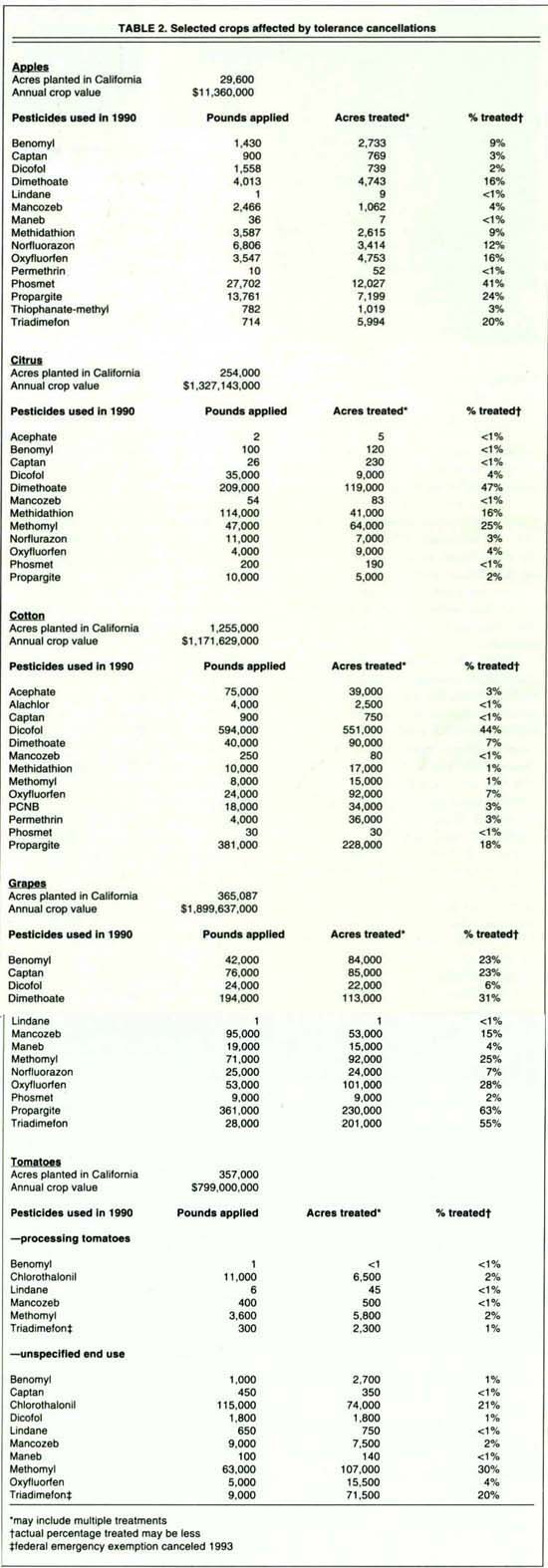
Many California growers have used some of these pesticides in their pest management programs. We have evaluated five crops to determine the extent to which growers have used the endangered pesticides in 1990. The crops are apples, citrus, cotton, grapes and tomatoes. Acreages, crop values and percentages of crops treated were determined (table 2). Some of the pesticides are not used in California, some are applied only to a minute fraction of the total crop, while a few are used extensively.
Residue testing shows that about 99% of agricultural products delivered to market have residues within legally acceptable limits.
Impact of Delaney ruling
It is difficult to predict the potential impact on the state's agricultural production if these pesticides are removed from the market since various alternative pest management chemicals or techniques may be adopted in response. Where alternatives are not available, production of specific commodities may be jeopardized.
For example, tomato growers may face serious losses from pesticide cancellations if the Delaney Clause is enforced according to EPA's policy proposal. The tomato crop, valued at $800 million annually, is grown on 357,000 acres of farmland in California. Both plants and fruit are attacked by insect, mite, nematode and fungus pests. Weeds are also serious problems in tomato fields.
California's pesticide use reports on tomatoes for 1990 show that one insecticide, methomyl, and two fungicides, chlorothalonil and mancozeb, are very important and were applied to a significant percentage of the state's tomatoes (table 2). The three pesticides could be lost under the EPA's proposed implementation of the Delaney Clause.
There are many alternatives to methomyl for insect pest management on tomatoes. There are a few chemical alternatives for chlorothalonil and mancozeb. Copper is an alternative control for early blight, late blight and blackmold, but copper is less effective than chlorothalonil and mancozeb. Triadimefon was used for powdery mildew in 1990, but it is no longer available for tomatoes in California because of a federal action canceling the federal emergency exemption from registration. Thus, tomato growers will have only one truly effective chemical, myclobutanil, for managing powdery mildew. (Sulfur is available, but it is much less effective.) For late blight, metalaxyl plus copper may be useful. In general, tomato growers will have to use less effective, less reliable and more costly cultural controls; their crop will be much more susceptible to losses from these diseases, especially following inclement weather. Fortunately, California does not usually receive rainfall during much of the growing season, so the crop will only be at risk in years with unusually wet growing seasons.
Delaney paradox
The paradox results from two sections of the Federal Food Drug and Cosmetic Act. Section 408 regulates pesticide residues on raw agricultural commodities and requires EPA to evaluate and consider both risks and benefits should the pesticide be registered. Section 409 regulates pesticide residues in processed agricultural commodites and does not allow risk and benefit evalution. It simply requires that residues be “safe.” Only risks can be evaluated. Section 409 includes the Delaney Clause, which prohibits food additives that have been shown to cause cancer in humans or animals.
If any statistically significant increase in cancers is found in animal tests, the law prohibits the residues in processed food. Paradoxically, the laws do not prohibit the same residues in the raw agricultural products that reach the market.
EPA's coordination policy: Because of the Delaney paradox, EPA developed a policy for coordinated tolerance setting that prohibits establishing a raw agricultural commodity tolerance if a tolerance on processed food is prohibited under Section 409.
However, this policy was abridged in the recent past when EPA carried out the Section 409 (Delaney Clause) provisions on the basis of the de minimus principle (see below). The use of the de minimus principle has now been overturned (see “Delaney update” opposite).
De minimis principle: Under this principle an administrative agency can avoid applying the terms of a law literally when doing so would lead to scientifically unsound results. Since the risks from certain pesticide residues are extremely low, even using worst-case extrapolation procedures, they have been considered to be “negligible” by EPA.
Negligible risk: Usually defined as the potential to cause one additional hypothetical cancer in a million people. Using the negligible risk criterion, EPA set tolerances on both raw and processed food for a few pesticides that had induced cancer in laboratory animal tests. —Editor
Canceling tolerances for fungicides chlorothalonil and mancozeb will leave tomato growers with few effective options for controlling powdery mildew (above), and blackmold (below).
Many pesticides have been canceled in the past, but usually cancellations have applied to only one chemical at a time. Generally there have been alternative chemicals or pest management techniques to replace the losses. Developing environmentally sound, efficacious and economical pest management methods is difficult, time consuming and expensive. Incremental pesticide losses allow agricultural technology to develop in response to pest management needs as they evolve. If the EPA adopts a coordination policy that results in the sudden loss of many widely used pesticides, it will be difficult to identify or develop alternatives before severe production problems result. Regional economies based on agriculture could suffer the consequences.
Pest management alternatives
It will be a challenge to avoid problems from future pesticide cancellations, but we are not without resources. Growers may face major pesticide losses because of changing federal interpretation of the Delaney Clause, registration losses under state or federal laws or from manufacturers' business decisions to remove products from the market. For various reasons, some pesticides will become unavailable. Growers must then determine the best alternative for managing their pest problems.
UC's Division of Agriculture and Natural Resources has a data base of scientifically valid pest management guidelines developed by faculty researchers, Cooperative Extension specialists and farm advisors. The information has been peer reviewed and edited, and it is updated frequently. Guidelines have been completed for 27 crop categories (table 3).
A typical UC pest management guideline describes the crop, the pest, the damage, possible biological controls and potential and known resistance problems. It also provides information on monitoring techniques, economic thresholds if any are available, treatment timing, comments on effective control and information on spot treatments. Finally, each guideline gives specific instructions on applying pesticides to ensure effective control.
As it has in the past, the search for reliable alternative pest management methods will continue to challenge UC's pest management programs for many years to come. The university will be called upon to support IPM activities, particularly for those minor crops most affected by cancellations of these pesticide registrations.
In light of future pesticide cancellations, UC researchers and growers are working to develop alternative methods of managing pest problems. Above, leaf removal is a cultural method of controlling fungal diseases such as Botrytis bunch rot.
Predatory insects, like the Metaseiulus occidentalis shown attacking a twospotted spider mite and its eggs (left), also give growers some options.
Delaney update
The 1958 Delaney Clause, contained in the Federal Food, Drug and Cosmetic Act (FFDCA), prohibits food additives in any amount if they have been shown to cause cancer in animals or humans. Since the law was written, evolving technology has enabled scientists to detect the presence of pesticides at much lower levels than before, routinely at 1 or 2 parts per billion. Taking this into account, the Environmental Protection Agency began enforcing the law on a de minimus basis, substituting a standard of negligible risk for one of zero risk (see opposite).
However, on February 25, 1991, the Natural Resources Defense Council, AFL-CIO and others filed suit in the Ninth Circuit Court of Appeals in San Francisco challenging EPA's interpretation of the Delaney Clause. On July 8, 1992, the federal court ruled that the Delaney Clause does not allow for such latitude in enforcement. On February 22, 1993, the Supreme Court denied a petition to review the Circuit Court's decision.
The EPA has asked Congress to update the pesticide regulations by this summer. Currently Congress is considering two bills — HR 1627 sponsored by Richard Lehman(D-CA), Thomas Bliley (D-GA) and Roy Rowland (R-VA) and S 331/HR 872 introduced by Edward Kennedy (D-MA) and Henry Waxman (D-CA) — to amend the FFDCA.
The Clinton Administration also has a pesticide safety reform package, which is expected to be submitted to Congress in the form of a bill in January. (For discussion of bills, see page 29 .) —Editor