All Issues
Sidebar: Debate simmers: Should carcinogens be tested at maximum tolerated dose?
Publication Information
California Agriculture 48(1):28-28.
Published January 01, 1994
PDF | Citation | Permissions
Full text
When chemicals are tested in laboratory animals to determine if they can cause cancer, they are tested at the maximum tolerated dose (MTD). The MTD is defined as the highest dose that can be administered to animals on a long-term basis that does not reduce their lifespans from causes other than cancer. MTDs are frequently much higher than the doses of chemicals that humans receive from environmental exposure. When chemicals are tested at such high doses, they can elicit physiologic alterations, including increased rates of cell proliferation, that would not be expected to occur at the lower doses to which humans are generally exposed. For example, a chemical that causes DNA damage will magnify the effects of that damage if its toxicity at high doses leads to increased cell proliferation. In this case, the chemical will appear to be a more potent carcinogen at high doses than it would be if it were tested at low doses.
The potential discrepancy between mechanisms of carcinogenesis operative at high doses and those operative at lower doses raises questions regarding the relevance of carcinogenicity testing at the MTD. Are chemicals identified as carcinogens only at high doses relevant to human exposure? Without some knowledge of the biologic mechanisms by which a chemical induces cancer at high doses, this is a difficult question to answer.
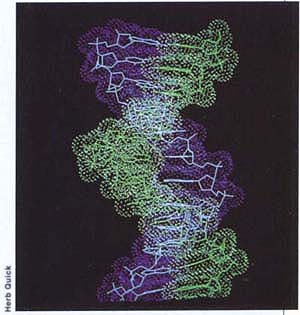
Cell proliferation plays a critical role in the development of cancer. When a cancer-causing agent reacts with a site on a cell's DNA (or genetic material), the cell may or may not be able to repair itself. If not, the DNA may be altered such that when it is copied prior to cell division, its sequence is changed permanently in the new cell. This change is called a mutation. If the mutation occurs at a site that is critical for controlling the cell's rate of proliferation or differentiation, the cell may be transformed into a cancer cell, which in turn can proliferate further and appear as a tumor. Increasing the rate of cell proliferation can reduce the time that a cell has to repair damage to DNA and thereby increase the likelihood that mutations will occur.
In general, the rate at which a cell proliferates can be increased in one of two ways. Some chemicals, called mitogens, can increase cell division by interacting with special receptors on a cell's surface that send signals to the sites on DNA that control proliferation. Other chemicals can be toxic to a group of cells, and increased cell division occurs as a consequence of toxicity as the remaining cells attempt to replace those that were killed.
The specificity and sensitivity of carcinogenicity tests in animals are important considerations for evaluating whether the results of animal tests can be used to predict a chemical's behavior in humans. “Sensitivity” refers to the ability of animal tests to detect true human carcinogens, and “specificity” refers to their ability to avoid identifying chemicals as human carcinogens when they are not. The sensitivity of animal tests appears to be high; all of the known human carcinogens tested have been positive in at least one animal species, although the data for target organs are often inconsistent. The specificity of animal tests cannot be evaluated at present, however, because available data on human noncarcinogens are insufficient to make comparisons. In the absence of information to the contrary, the assumption is made by regulatory agencies that evidence supporting or refuting carcinogenic activity in animal studies should be considered applicable to humans until better information is obtained.
Computer-generated image of DNA; its reaction with a cancer-causing agent may lead to a mutation if the cell is unable to repair itself.
There is controversy in the scientific community regarding the use of the MTD. One view is that the MTD should continue to be used to screen chemicals for potential carcinogenicity because using lower doses could sacrifice the sensitivity of animal tests; that is, fewer chemicals would be identified as carcinogens at lower doses, and a potential human carcinogen could be missed. The other view is that instead of using the MTD, the highest dose tested should be that which elicits effects comparable to those expected if actual human exposure is taken into account. This would increase the specificity of animal tests. Choosing the dose that is most relevant to humans is likely to be difficult without knowledge of each chemical's biologic mechanisms of action, however. Understanding a chemical's biologic mechanisms of action requires more extensive testing than is generally performed at present.